Practice Essentials
Heterotopic ossification (HO) is the abnormal formation of true bone within extraskeletal soft tissues. Classically, many diseases sharing this common feature were lumped into the category myositis ossificans; however, the latter term has fallen into disfavor because primary muscle inflammation is not a necessary precursor for such ossification, and the ossification does not always occur in muscle tissue (frequently showing a predilection, instead, for fascia, tendons, and other mesenchymal soft tissues). The term heterotopic ossification has largely replaced myositis ossificans in the literature. [1, 2, 3, 4]
(Heterotopic ossification is displayed in the images below.)
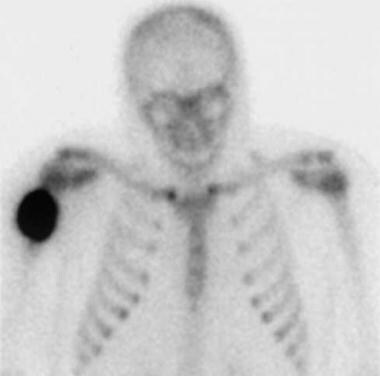 Delayed image from a technetium-99m (99mTc) methylene diphosphonate bone scan (performed at the time of the first radiograph) demonstrates marked soft-tissue uptake of tracer.
Delayed image from a technetium-99m (99mTc) methylene diphosphonate bone scan (performed at the time of the first radiograph) demonstrates marked soft-tissue uptake of tracer.
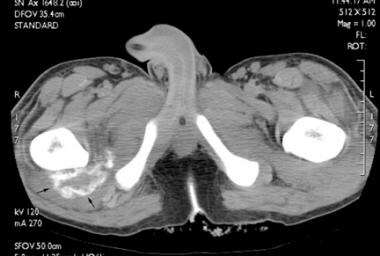
 Noncontrast, axial computed tomography (CT) scan through the proximal thighs in a paraplegic patient with long-standing spinal cord injury. Mature heterotopic ossification surrounds the femoral shafts bilaterally, nearly obscuring the right femur (black arrowhead). A large decubitus ulcer overlies the ossification posteriorly (white arrows).
Noncontrast, axial computed tomography (CT) scan through the proximal thighs in a paraplegic patient with long-standing spinal cord injury. Mature heterotopic ossification surrounds the femoral shafts bilaterally, nearly obscuring the right femur (black arrowhead). A large decubitus ulcer overlies the ossification posteriorly (white arrows).
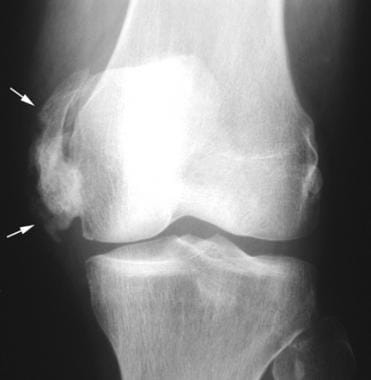 Anteroposterior radiograph of the left knee in a patient with traumatic brain injury. Mature heterotopic ossification surrounds the medial femoral condyle, with a solid peripheral cortex (arrows).
Anteroposterior radiograph of the left knee in a patient with traumatic brain injury. Mature heterotopic ossification surrounds the medial femoral condyle, with a solid peripheral cortex (arrows).
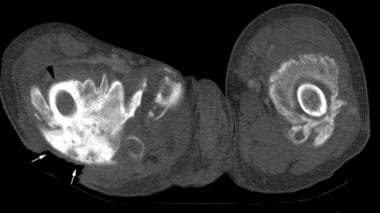
 Axial computed tomography (CT) scan in a male patient with lower extremity swelling 8 weeks after a spinal cord injury. Peripheral mineralization (arrows) is seen in immature heterotopic ossification within the quadratus femoris muscle.
Axial computed tomography (CT) scan in a male patient with lower extremity swelling 8 weeks after a spinal cord injury. Peripheral mineralization (arrows) is seen in immature heterotopic ossification within the quadratus femoris muscle.
Traditionally, various forms of HO have been classified according to the clinical setting and location of the lesions, as well as according to whether the lesions are progressive or isolated in occurrence.
The varieties of HO are as follows:
-
Myositis ossificans traumatica: HO occurring after recalled trauma, such as blunt injury, surgery, or burns
-
Nontraumatic myositis ossificans: HO that occurs when no inciting trauma can be identified
-
Panniculitis ossificans: HO confined to subcutaneous fat
-
Rider's bones: HO found in the adductor muscles
-
Shooter's bones: HO located in the deltoid muscle
Genetic HO occurs in individuals with inherited conditions such as fibrodysplasia ossificans progressiva and progressive osseous heteroplasia. [5]
Fibrodysplasia ossificans progressiva (FOP), or Munchmeyer disease, is an autosomal dominant, severely disabling condition that results in progressive ossification of fascial planes, muscles, tendons, and ligaments. Congenital malformation of the great toes also is associated with FOP. HO is a feature of several other diseases as well, including Albright hereditary osteodystrophy, progressive osseous heteroplasia, and primary osteoma cutis.
Traumatic HO occurs in response to injuries such as acetabular fractures, fractures and joint dislocations, blast injuries, burns, combat, amputee injuries, and muscle trauma. Neurogenic HO occurs in response to traumatic brain injury (TBI) and spinal cord injuries. A strong association exists between HO and spinal cord injury, with lesions occurring at multiple sites and showing a strong propensity to recur. [3] Similarly, periarticular HO is seen in patients with traumatic brain injury, with the extent and functional severity of the HO directly related to the severity of the intracranial injury. [6]
Many other causes of neurologic compromise, including tetanus, poliomyelitis, Guillain-Barré syndrome, and prolonged pharmacologic paralysis during mechanical ventilation, also have been associated with HO formation.
Preferred examination
Radiography is the preferred method of initial assessment for virtually all musculoskeletal conditions, including HO. Given their relatively low expense, radiographs should be obtained first (even if other imaging modalities are planned) in order to assess the extent of known HO. [7, 8]
Bone scanning is the method of choice for earliest detection and, once the diagnosis is established, for assessing the maturity of a known lesion. Bone scans are typically positive more than 2 weeks before radiographic evidence of mineralization is seen, with some authors reporting positive findings on bone scan 4-6 weeks earlier than on radiographs.
Ultrasonography may have a role as a screening tool in the hip region after spinal cord injury; ultrasonograms may be obtained during a DVT screening examination.
CT may be used to delineate the area of bone formation in preparation for surgery. MRI may also be used but is not cost-effective unless bone encompasses neurologic structures. [9]
Radiographs cannot detect the mineralization of HO during the first 1-2 weeks after the inciting trauma or the onset of symptoms. However, radiographs are recommended in all patients with suggested HO to assess underlying bone pathology and exclude other pathology.
Neither radiography nor computed tomography (CT) scanning should be performed in the pelvic region during pregnancy because of radiation exposure to the fetus, unless the risks of radiation exposure are outweighed by the need for a timely diagnosis. [10] Nuclear medicine bone scanning also involves exposure of the fetus to radiation, regardless of the anatomic site being imaged, and should, if possible, be postponed until after delivery.
Ultrasonography is an operator-dependent modality. Proper evaluation of soft-tissue masses, including HO, requires considerable training and experience, available at some centers in the United States and, more commonly, in Europe. Radiologists who are less familiar with musculoskeletal ultrasonographic images may be more comfortable using CT scanning or nuclear medicine modalities to make the diagnosis of early HO.
The Brooker classification is a commonly used tool to characterize the extent of heterotopic ossification. It divides the extent of HO formation into 4 grades [11] :
-
Grade I describes small separate small foci of bony islands within the soft tissues about the hip.
-
Grade II consists of ossifications projecting from the proximal femur or pelvis with at least 1 cm between opposing bone surfaces.
-
Grade III includes bone spurs originating from the proximal femur or pelvis with a separation distance of less than 1 cm.
-
Grade IV describes complete ossification bridging between the proximal femur and pelvis.
Limitatins with the Brooker classification include superimposing structures, variations in radiographic technique, and unaccountability for differences in volumetric measurements and arthroplasty lengths. [11]
Medical/legal pitfalls
Failure to distinguish heterotopic ossification from an osteoblastic neoplasm, such as osteosarcoma, is a pitfall.
Aggressive imaging findings may lead to a percutaneous biopsy, which can yield a sample with high cellular activity and new bone formation; these characteristics can potentially be confusing to the pathologist. An open biopsy may fare no better; it can exacerbate the severity of HO or, if the biopsy follows an early resection, cause a recurrence of the condition. Biopsy is contraindicated in patients with an established diagnosis of fibrodysplasia ossificans progressiva.
Another pitfall is the failure to use a conservative approach to follow-up radiologic imaging if developing HO is in the differential diagnosis for a soft-tissue mass. Films can be obtained over a period of a few months at 2- to 4-week intervals to assess for the maturation of the osseous elements and increasing circumscription of the lesion (as opposed to the characteristics of an aggressive neoplasm's progression).
Radiography
A soft tissue mass is the earliest radiographic finding of heterotopic ossification (HO), although a mass is often overlooked at sites such as the hip and thigh. Mineralization of the osteoid lesional content can be seen as early as 10-14 days after the inciting trauma. A peripheral zone of early mineralization is the most common pattern, although many lesions have a less organized appearance. Typically, HO associated with fibrodysplasia ossificans progressiva first mineralizes centrally. If left untreated, mature cortical bone ultimately results.
Peripheral ossification of a soft tissue mass at the site of recent trauma is consistent with HO. Performing a biopsy to exclude a neoplasm should be avoided. Instead, delayed radiographs should be performed in 4 weeks to confirm maturation of the lesion into typical HO.
False positives can be caused by avulsion fracture fragments, osteochondral bodies within a distended joint capsule, nonosseous soft tissue calcification (which can resemble amorphous, early mineralization of woven bone), and osteosarcoma. False negatives are common, most often occurring in lesions in which it is too early for mineralization to be seen radiographically or at sites that have been obscured by generous, overlying soft tissues or by normal bone. Sensitivity and specificity increase with time as lesions display more mature, dense ossification.
(See the radiographs of heterotopic ossification below.)
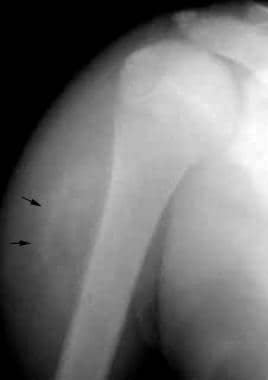 Anteroposterior radiograph of the right shoulder in a young man with a painful mass following recent blunt trauma. Soft-tissue swelling is present. A linear band of early mineralization (arrows) is faintly visible.
Anteroposterior radiograph of the right shoulder in a young man with a painful mass following recent blunt trauma. Soft-tissue swelling is present. A linear band of early mineralization (arrows) is faintly visible.
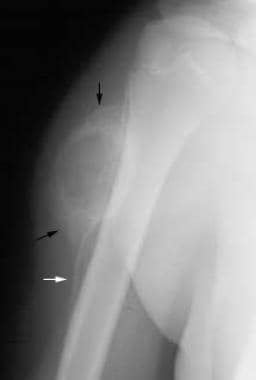 Anteroposterior radiograph obtained 5 weeks after the initial film demonstrates that maturing peripheral bone (black arrows) has replaced the initial mineralization. The mineralization pattern roughly corresponds with the peripheral low signal seen on magnetic resonance images. A solid periosteal reaction extends distally from the lesion (white arrow).
Anteroposterior radiograph obtained 5 weeks after the initial film demonstrates that maturing peripheral bone (black arrows) has replaced the initial mineralization. The mineralization pattern roughly corresponds with the peripheral low signal seen on magnetic resonance images. A solid periosteal reaction extends distally from the lesion (white arrow).
 Anteroposterior radiograph of the left knee in a patient with traumatic brain injury. Mature heterotopic ossification surrounds the medial femoral condyle, with a solid peripheral cortex (arrows).
Anteroposterior radiograph of the left knee in a patient with traumatic brain injury. Mature heterotopic ossification surrounds the medial femoral condyle, with a solid peripheral cortex (arrows).
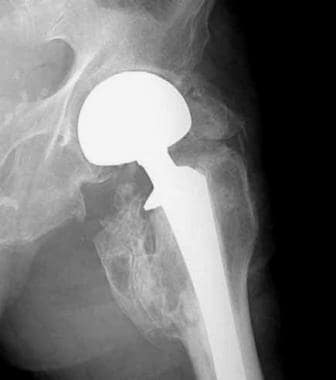 Attempted frog-leg, lateral radiograph of the left hip. Mature heterotopic ossification (HO) surrounds the greater and lesser trochanters of the femur in this patient, who had a bipolar hip prosthesis placed 2 months earlier. The large amount of HO resulted in a significantly reduced range of motion.
Attempted frog-leg, lateral radiograph of the left hip. Mature heterotopic ossification (HO) surrounds the greater and lesser trochanters of the femur in this patient, who had a bipolar hip prosthesis placed 2 months earlier. The large amount of HO resulted in a significantly reduced range of motion.
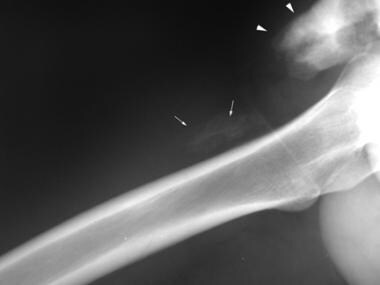
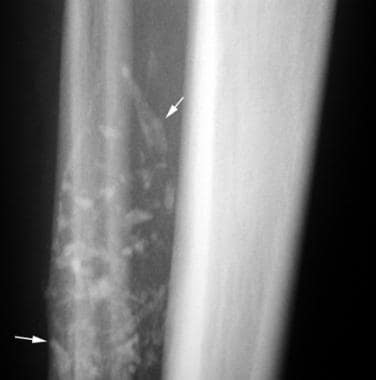 Magnified view of an anteroposterior radiograph of the right tibia in a patient with a palpable anterior mass of the lower leg and a remote history of trauma. Dystrophic-appearing calcifications project over the interosseous membrane and fibular shaft.
Magnified view of an anteroposterior radiograph of the right tibia in a patient with a palpable anterior mass of the lower leg and a remote history of trauma. Dystrophic-appearing calcifications project over the interosseous membrane and fibular shaft.
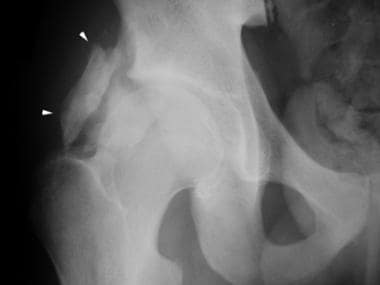 Anteroposterior radiograph of the right hip in a 16-year-old boy who had suffered trauma to the hip 2 years previously. The patient is currently experiencing hip pain. Mature heterotopic ossification (arrowheads) projects over and lateral to the femoral head.
Anteroposterior radiograph of the right hip in a 16-year-old boy who had suffered trauma to the hip 2 years previously. The patient is currently experiencing hip pain. Mature heterotopic ossification (arrowheads) projects over and lateral to the femoral head.
 Lateral view of the right hip. Distal to the mature heterotopic ossification (HO) seen on the anteroposterior view (arrowheads) is a subtle area of early mineralization (arrows) consistent with early HO.
Lateral view of the right hip. Distal to the mature heterotopic ossification (HO) seen on the anteroposterior view (arrowheads) is a subtle area of early mineralization (arrows) consistent with early HO.
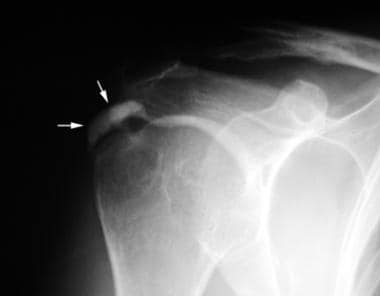 Anteroposterior radiograph performed in external rotation of the right shoulder in a 62-year-old female patient. Amorphous calcification is seen superolateral to the humeral head (arrows). The characteristics of this calcification are not indicative of heterotopic ossification (HO); the mature cortical or trabecular structures seen in mature HO are not found, nor are the well-defined margins that are consistent with the immature woven bone of early HO.
Anteroposterior radiograph performed in external rotation of the right shoulder in a 62-year-old female patient. Amorphous calcification is seen superolateral to the humeral head (arrows). The characteristics of this calcification are not indicative of heterotopic ossification (HO); the mature cortical or trabecular structures seen in mature HO are not found, nor are the well-defined margins that are consistent with the immature woven bone of early HO.
Computed Tomography
The earliest CT scan findings include a low-attenuation soft tissue mass or an enlarged muscle belly, occasionally with indistinct, adjacent soft-tissue planes (see the images below). [12, 13] Faint, immature bone mineralization becomes evident earlier than it does on radiographs, as lesions mature. With further maturation, lesions often demonstrate zonal mineralization with a mature peripheral cortex and may have central, fat-containing marrow elements. Abnormal, low-attenuation findings may persist in the soft tissues surrounding the mature cortex for months to years after the disease's onset. [14, 15, 16]
CT scans demonstrate high specificity for HO when the typical zonal pattern of peripheral mineralization is present. [17] Earlier in the course of HO evolution, when the lesion appears as an enhancing mass with disorganized or absent mineralization, findings are nonspecific. A repeat CT scan after several weeks of further maturation may help resolve the diagnostic dilemma.
Many masslike conditions, including the presence of an inflammatory mass or a soft tissue neoplasm, can mimic the early appearance of HO.
 Noncontrast, axial computed tomography (CT) scan through the proximal thighs in a paraplegic patient with long-standing spinal cord injury. Mature heterotopic ossification surrounds the femoral shafts bilaterally, nearly obscuring the right femur (black arrowhead). A large decubitus ulcer overlies the ossification posteriorly (white arrows).
Noncontrast, axial computed tomography (CT) scan through the proximal thighs in a paraplegic patient with long-standing spinal cord injury. Mature heterotopic ossification surrounds the femoral shafts bilaterally, nearly obscuring the right femur (black arrowhead). A large decubitus ulcer overlies the ossification posteriorly (white arrows).
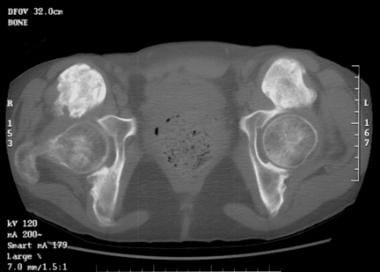 Noncontrast, axial computed tomography (CT) scan through the hips of a young woman with long-standing spinal cord injury. Large masses of mature heterotopic ossification replace the iliopsoas muscles anterior to both femoral heads.
Noncontrast, axial computed tomography (CT) scan through the hips of a young woman with long-standing spinal cord injury. Large masses of mature heterotopic ossification replace the iliopsoas muscles anterior to both femoral heads.
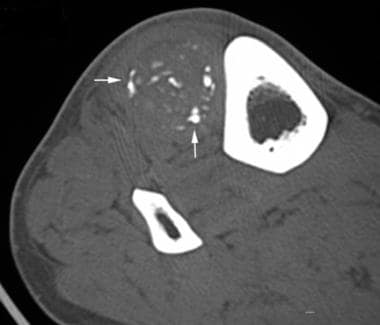 Noncontrast, axial computed tomography (CT) scan through a palpable mass demonstrates dystrophic-appearing calcifications scattered throughout a soft-tissue mass in the anterior compartment. An open biopsy revealed heterotopic ossification (HO). While mature HO typically has lamellar peripheral bone in a zonal distribution, bizarre patterns of mineralization also can occur.
Noncontrast, axial computed tomography (CT) scan through a palpable mass demonstrates dystrophic-appearing calcifications scattered throughout a soft-tissue mass in the anterior compartment. An open biopsy revealed heterotopic ossification (HO). While mature HO typically has lamellar peripheral bone in a zonal distribution, bizarre patterns of mineralization also can occur.
 Axial computed tomography (CT) scan in a male patient with lower extremity swelling 8 weeks after a spinal cord injury. Peripheral mineralization (arrows) is seen in immature heterotopic ossification within the quadratus femoris muscle.
Axial computed tomography (CT) scan in a male patient with lower extremity swelling 8 weeks after a spinal cord injury. Peripheral mineralization (arrows) is seen in immature heterotopic ossification within the quadratus femoris muscle.
Magnetic Resonance Imaging
Magnetic resonance imaging (MRI) is not routinely employed for the evaluation of heterotopic ossification (HO) once the diagnosis is established. [18, 19] Cases are most often encountered when MRI is required to assess a mass that has been detected clinically. HO may be seen incidentally in patients at risk, such as paraplegics, in whom it may be found while they are undergoing assessment for pelvic osteomyelitis. [10]
As on CT scans, early lesions have a very nonspecific appearance on magnetic resonance images. [20] With time, the development of a zonal pattern of maturation may suggest HO, although the appearance is often misleading. Correlation with conventional radiographs is strongly recommended to confirm the diagnosis. On magnetic resonance images, as on CT scans, many masslike conditions, including infectious, inflammatory, and neoplastic disorders, can have an appearance similar to that of HO.
(See MRIs of heterotopic ossification below.)
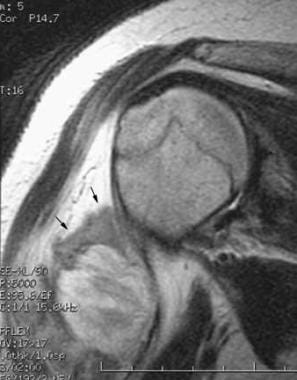 Coronal, oblique, T2-weighted, fast spin-echo magnetic resonance image of the right shoulder obtained on the same day as the initial radiograph. A mass in the soft tissues lateral to the proximal right humerus has a central, heterogeneous, high T2 signal with a surrounding zone of lower signal intensity (arrows).
Coronal, oblique, T2-weighted, fast spin-echo magnetic resonance image of the right shoulder obtained on the same day as the initial radiograph. A mass in the soft tissues lateral to the proximal right humerus has a central, heterogeneous, high T2 signal with a surrounding zone of lower signal intensity (arrows).
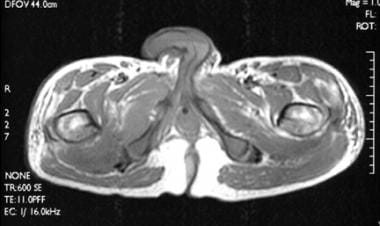 Corresponding noncontrast, axial, T1-weighted image from a magnetic resonance imaging (MRI) scan performed on the same day as the axial computed tomography (CT) scan demonstrates a masslike enlargement of the right quadratus femoris.
Corresponding noncontrast, axial, T1-weighted image from a magnetic resonance imaging (MRI) scan performed on the same day as the axial computed tomography (CT) scan demonstrates a masslike enlargement of the right quadratus femoris.
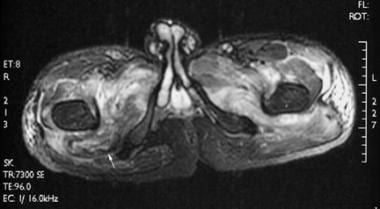 Axial, T2-weighted, fast spin-echo, fat-suppressed magnetic resonance image. Extensive edema is present bilaterally in several muscle groups. A high T2 signal alone is a nonspecific finding in patients with spinal cord injury, potentially heralding a site of heterotopic ossification development but often resolving spontaneously. A low signal seen peripherally in the right quadratus femoris (arrow) corresponds to the mineralization observed on computed tomography (CT) scan.
Axial, T2-weighted, fast spin-echo, fat-suppressed magnetic resonance image. Extensive edema is present bilaterally in several muscle groups. A high T2 signal alone is a nonspecific finding in patients with spinal cord injury, potentially heralding a site of heterotopic ossification development but often resolving spontaneously. A low signal seen peripherally in the right quadratus femoris (arrow) corresponds to the mineralization observed on computed tomography (CT) scan.
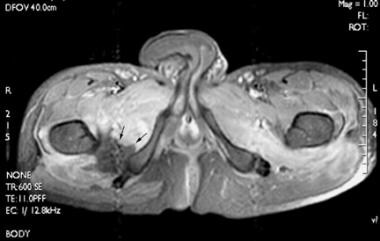 Axial, spin-echo, T1-weighted, fat-suppressed magnetic resonance image following intravenous gadolinium administration. In addition to extensive abnormal enhancement surrounding both hips, an unusual nonenhancing area (arrows) is noted anteromedial to heterotopic ossification (HO) demonstrated on computed tomography (CT) scan. The corresponding T2-weighted image does not show a discrete fluid collection in the region. A similar pattern has been seen in association with HO in other patients with spinal cord injuries.
Axial, spin-echo, T1-weighted, fat-suppressed magnetic resonance image following intravenous gadolinium administration. In addition to extensive abnormal enhancement surrounding both hips, an unusual nonenhancing area (arrows) is noted anteromedial to heterotopic ossification (HO) demonstrated on computed tomography (CT) scan. The corresponding T2-weighted image does not show a discrete fluid collection in the region. A similar pattern has been seen in association with HO in other patients with spinal cord injuries.
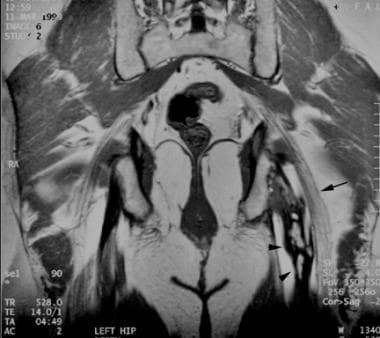 Coronal, T1-weighted magnetic resonance image through the posterior pelvis in a patient with left-sided sciatica. An elongated mass of mature heterotopic ossification (arrowheads) with a peripheral low signal and a central fat signal replaces the proximal hamstrings parallel and adjacent to the sciatic nerve (arrow).
Coronal, T1-weighted magnetic resonance image through the posterior pelvis in a patient with left-sided sciatica. An elongated mass of mature heterotopic ossification (arrowheads) with a peripheral low signal and a central fat signal replaces the proximal hamstrings parallel and adjacent to the sciatic nerve (arrow).
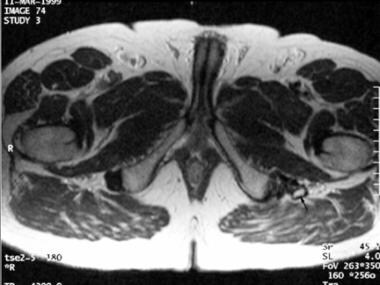 Axial, T2-weighted, fast spin-echo magnetic resonance image through the lower pelvis demonstrates the marrow fat (arrow) within the mature heterotopic ossification (HO) that replaces the semitendinosus and biceps femoris tendons. HO in this region results from a prior ischial tuberosity avulsion injury and can be symptomatic when it impinges on the adjacent sciatic nerve.
Axial, T2-weighted, fast spin-echo magnetic resonance image through the lower pelvis demonstrates the marrow fat (arrow) within the mature heterotopic ossification (HO) that replaces the semitendinosus and biceps femoris tendons. HO in this region results from a prior ischial tuberosity avulsion injury and can be symptomatic when it impinges on the adjacent sciatic nerve.
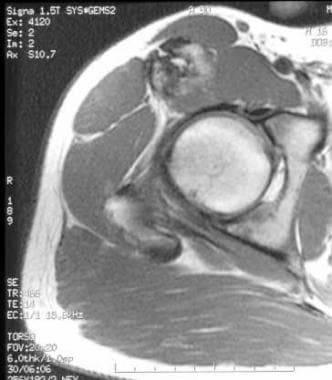 Axial, T1-weighted magnetic resonance image through the proximal, mature heterotopic ossification demonstrates a heterogeneous mass—replacing the rectus femoris and a portion of the iliopsoas—with a central fat signal anterior to the femoral head.
Axial, T1-weighted magnetic resonance image through the proximal, mature heterotopic ossification demonstrates a heterogeneous mass—replacing the rectus femoris and a portion of the iliopsoas—with a central fat signal anterior to the femoral head.
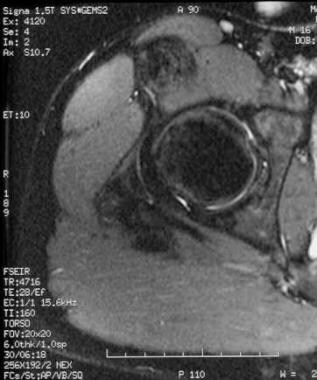 Axial, T2-weighted, short tau inversion recovery (STIR) image demonstrates a low signal and shows no appreciable edema.
Axial, T2-weighted, short tau inversion recovery (STIR) image demonstrates a low signal and shows no appreciable edema.
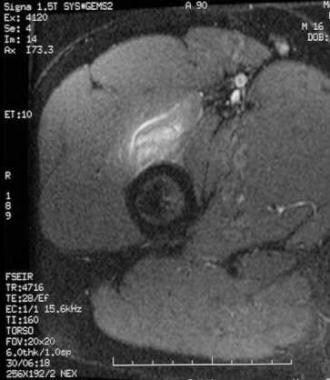 Axial, T2-weighted, short tau inversion recovery (STIR) image through the more distal area of early mineralization reveals prominent soft-tissue edema consistent with early, immature heterotopic ossification.
Axial, T2-weighted, short tau inversion recovery (STIR) image through the more distal area of early mineralization reveals prominent soft-tissue edema consistent with early, immature heterotopic ossification.
Early HO lesions demonstrate a heterogeneous, high T2 signal and a masslike enlargement of affected tissues. [21] A low-signal-intensity rim may be seen. Frequently, an extensive, ill-defined, surrounding, high T2 signal is noted. Intravenous gadolinium administration results in early, intense, heterogeneous enhancement of lesions.
Delayed imaging that takes place weeks to months after onset demonstrates a low-signal rim corresponding to maturing cortical bone on radiographs, with persistence of the heterogeneous, high T2 signal lesional content and extensive surrounding edema.
Several months after HO's onset, imaging characteristics reveal decreasing edema within and surrounding the lesion. High T1 and T2 signals that are isointense to fat develop centrally, probably representing marrow fat. Late lesions typically do not enhance.
Gadolinium-based contrast agents have been linked to the development of nephrogenic systemic fibrosis (NSF) or nephrogenic fibrosing dermopathy (NFD). The disease has occurred in patients with moderate to end-stage renal disease after being given a gadolinium-based contrast agent to enhance MRI or magnetic resonance angiography (MRA) scans. NSF/NFD is a debilitating and sometimes fatal disease. Characteristics include red or dark patches on the skin; burning, itching, swelling, hardening, and tightening of the skin; yellow spots on the whites of the eyes; joint stiffness with trouble moving or straightening the arms, hands, legs, or feet; pain deep in the hip bones or ribs; and muscle weakness.
Ultrasonography
Ultrasonography is not generally used to assess heterotopic ossification once the diagnosis is established. However, because of the overlap of clinical presentation with deep venous thrombosis, combined with the widespread use of ultrasonography to screen for DVT, ultrasonographers can expect to encounter HO in the lower extremities of patients at risk. [22, 23] Some authors have proposed the use of ultrasonography as a simultaneous screening tool for HO and DVT in patients with spinal cord injury (see the image below).
As with HO findings on magnetic resonance images, those on ultrasonograms should be confirmed with radiographs and followed for the typical maturation pattern of HO. Early in the disorganized, preradiographic phase, HO can have an appearance similar to that of a variety of posttraumatic, inflammatory, and neoplastic conditions.
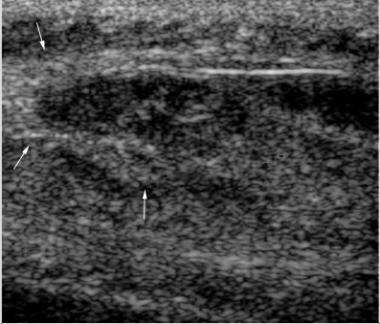 Longitudinal ultrasonogram of the medial thigh in a 20-year-old patient with paraplegia who has a hard, palpable mass. The heterogeneous, central, hypoechogenic mass is surrounded peripherally by more echogenic material (arrows). A core biopsy under ultrasonographic guidance revealed immature heterotopic ossification (HO). A computed tomography (CT) scan at the time did not demonstrate mineralization, although later radiographs showed typical HO. Courtesy of Dr Robert Lopez, Department of Radiology, University of Alabama at Birmingham.
Longitudinal ultrasonogram of the medial thigh in a 20-year-old patient with paraplegia who has a hard, palpable mass. The heterogeneous, central, hypoechogenic mass is surrounded peripherally by more echogenic material (arrows). A core biopsy under ultrasonographic guidance revealed immature heterotopic ossification (HO). A computed tomography (CT) scan at the time did not demonstrate mineralization, although later radiographs showed typical HO. Courtesy of Dr Robert Lopez, Department of Radiology, University of Alabama at Birmingham.
Within a few hours of the onset of clinical symptoms, ultrasonograms demonstrate a chaotic disruption of the normal lamellar structure of skeletal muscle, which can be seen up to 10-14 days before radiographic evidence of HO appears. Later, still in the preradiographic phase, a zonal, masslike pattern develops that is peripherally echogenic and centrally hypoechoic. Once early mineralization becomes radiographically visible, ultrasonograms demonstrate sheets or irregular clumps of echogenic material with acoustic shadowing. Mature lesions have the echogenicity and dense shadowing of cortical bone.
Nuclear Imaging
A 3-phase, radionuclide bone scan using technetium-99m (99mTc) methylene diphosphonate remains the criterion standard for detecting early heterotopic ossification. [24, 25] The examination consists of a radionuclide angiogram followed by a blood pool image over the suggestive site. A delayed image is obtained 2-3 hours later.
Bone scans are typically positive more than 2 weeks before radiographic evidence of mineralization is seen, with some authors reporting positive findings on bone scan 4-6 weeks earlier than on radiographs. In very early lesions, only the first-pass or blood pool image findings may be abnormal, reflecting the hypervascularity of early heterotopic ossification. Soft tissue uptake in the third phase subsequently develops within 1 week of the appearance of first-pass and second-pass abnormalities. Soft tissue uptake is considered diagnostic of HO in the appropriate clinical setting.
(See the images below.)
 Delayed image from a technetium-99m (99mTc) methylene diphosphonate bone scan (performed at the time of the first radiograph) demonstrates marked soft-tissue uptake of tracer.
Delayed image from a technetium-99m (99mTc) methylene diphosphonate bone scan (performed at the time of the first radiograph) demonstrates marked soft-tissue uptake of tracer.
 First-pass, or angiogram phase, of a technetium-99m (99mTc) methylene diphosphonate bone scan on a 16-year-old boy demonstrates no significant abnormality.
First-pass, or angiogram phase, of a technetium-99m (99mTc) methylene diphosphonate bone scan on a 16-year-old boy demonstrates no significant abnormality.
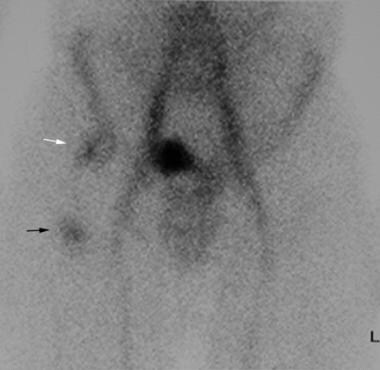 Second-pass, blood pool image demonstrates abnormal uptake in the right femoral head region (white arrow) corresponding to the mature heterotopic ossification (HO) seen radiographically. A second area of uptake over the proximal right femoral shaft (black arrow) correlates with the wispy mineralization of early HO, seen on the lateral view.
Second-pass, blood pool image demonstrates abnormal uptake in the right femoral head region (white arrow) corresponding to the mature heterotopic ossification (HO) seen radiographically. A second area of uptake over the proximal right femoral shaft (black arrow) correlates with the wispy mineralization of early HO, seen on the lateral view.
Gallium-67 uptake is commonly demonstrated and may potentially be confused with infection in patients with spinal cord injuries.
-
Anteroposterior radiograph of the right shoulder in a young man with a painful mass following recent blunt trauma. Soft-tissue swelling is present. A linear band of early mineralization (arrows) is faintly visible.
-
Delayed image from a technetium-99m (99mTc) methylene diphosphonate bone scan (performed at the time of the first radiograph) demonstrates marked soft-tissue uptake of tracer.
-
Coronal, oblique, T2-weighted, fast spin-echo magnetic resonance image of the right shoulder obtained on the same day as the initial radiograph. A mass in the soft tissues lateral to the proximal right humerus has a central, heterogeneous, high T2 signal with a surrounding zone of lower signal intensity (arrows).
-
Anteroposterior radiograph obtained 5 weeks after the initial film demonstrates that maturing peripheral bone (black arrows) has replaced the initial mineralization. The mineralization pattern roughly corresponds with the peripheral low signal seen on magnetic resonance images. A solid periosteal reaction extends distally from the lesion (white arrow).
-
Noncontrast, axial computed tomography (CT) scan through the proximal thighs in a paraplegic patient with long-standing spinal cord injury. Mature heterotopic ossification surrounds the femoral shafts bilaterally, nearly obscuring the right femur (black arrowhead). A large decubitus ulcer overlies the ossification posteriorly (white arrows).
-
Noncontrast, axial computed tomography (CT) scan through the hips of a young woman with long-standing spinal cord injury. Large masses of mature heterotopic ossification replace the iliopsoas muscles anterior to both femoral heads.
-
Anteroposterior radiograph of the left knee in a patient with traumatic brain injury. Mature heterotopic ossification surrounds the medial femoral condyle, with a solid peripheral cortex (arrows).
-
Anteroposterior radiograph of the right knee in a patient with traumatic brain injury demonstrates mature heterotopic ossification in the same distribution as on the left knee. The hips, shoulders, and elbows are more common sites of involvement in the setting of traumatic brain injury.
-
Longitudinal ultrasonogram of the medial thigh in a 20-year-old patient with paraplegia who has a hard, palpable mass. The heterogeneous, central, hypoechogenic mass is surrounded peripherally by more echogenic material (arrows). A core biopsy under ultrasonographic guidance revealed immature heterotopic ossification (HO). A computed tomography (CT) scan at the time did not demonstrate mineralization, although later radiographs showed typical HO. Courtesy of Dr Robert Lopez, Department of Radiology, University of Alabama at Birmingham.
-
Attempted frog-leg, lateral radiograph of the left hip. Mature heterotopic ossification (HO) surrounds the greater and lesser trochanters of the femur in this patient, who had a bipolar hip prosthesis placed 2 months earlier. The large amount of HO resulted in a significantly reduced range of motion.
-
Magnified view of an anteroposterior radiograph of the right tibia in a patient with a palpable anterior mass of the lower leg and a remote history of trauma. Dystrophic-appearing calcifications project over the interosseous membrane and fibular shaft.
-
Noncontrast, axial computed tomography (CT) scan through a palpable mass demonstrates dystrophic-appearing calcifications scattered throughout a soft-tissue mass in the anterior compartment. An open biopsy revealed heterotopic ossification (HO). While mature HO typically has lamellar peripheral bone in a zonal distribution, bizarre patterns of mineralization also can occur.
-
Axial computed tomography (CT) scan in a male patient with lower extremity swelling 8 weeks after a spinal cord injury. Peripheral mineralization (arrows) is seen in immature heterotopic ossification within the quadratus femoris muscle.
-
Corresponding noncontrast, axial, T1-weighted image from a magnetic resonance imaging (MRI) scan performed on the same day as the axial computed tomography (CT) scan demonstrates a masslike enlargement of the right quadratus femoris.
-
Axial, T2-weighted, fast spin-echo, fat-suppressed magnetic resonance image. Extensive edema is present bilaterally in several muscle groups. A high T2 signal alone is a nonspecific finding in patients with spinal cord injury, potentially heralding a site of heterotopic ossification development but often resolving spontaneously. A low signal seen peripherally in the right quadratus femoris (arrow) corresponds to the mineralization observed on computed tomography (CT) scan.
-
Axial, spin-echo, T1-weighted, fat-suppressed magnetic resonance image following intravenous gadolinium administration. In addition to extensive abnormal enhancement surrounding both hips, an unusual nonenhancing area (arrows) is noted anteromedial to heterotopic ossification (HO) demonstrated on computed tomography (CT) scan. The corresponding T2-weighted image does not show a discrete fluid collection in the region. A similar pattern has been seen in association with HO in other patients with spinal cord injuries.
-
Coronal, T1-weighted magnetic resonance image through the posterior pelvis in a patient with left-sided sciatica. An elongated mass of mature heterotopic ossification (arrowheads) with a peripheral low signal and a central fat signal replaces the proximal hamstrings parallel and adjacent to the sciatic nerve (arrow).
-
Axial, T2-weighted, fast spin-echo magnetic resonance image through the lower pelvis demonstrates the marrow fat (arrow) within the mature heterotopic ossification (HO) that replaces the semitendinosus and biceps femoris tendons. HO in this region results from a prior ischial tuberosity avulsion injury and can be symptomatic when it impinges on the adjacent sciatic nerve.
-
Anteroposterior radiograph of the right hip in a 16-year-old boy who had suffered trauma to the hip 2 years previously. The patient is currently experiencing hip pain. Mature heterotopic ossification (arrowheads) projects over and lateral to the femoral head.
-
Lateral view of the right hip. Distal to the mature heterotopic ossification (HO) seen on the anteroposterior view (arrowheads) is a subtle area of early mineralization (arrows) consistent with early HO.
-
First-pass, or angiogram phase, of a technetium-99m (99mTc) methylene diphosphonate bone scan on a 16-year-old boy demonstrates no significant abnormality.
-
Second-pass, blood pool image demonstrates abnormal uptake in the right femoral head region (white arrow) corresponding to the mature heterotopic ossification (HO) seen radiographically. A second area of uptake over the proximal right femoral shaft (black arrow) correlates with the wispy mineralization of early HO, seen on the lateral view.
-
Third-phase, delayed image also shows abnormal tracer uptake over the femoral head (arrowhead) and proximal femoral shaft (arrow).
-
Axial, T1-weighted magnetic resonance image through the proximal, mature heterotopic ossification demonstrates a heterogeneous mass—replacing the rectus femoris and a portion of the iliopsoas—with a central fat signal anterior to the femoral head.
-
Axial, T2-weighted, short tau inversion recovery (STIR) image demonstrates a low signal and shows no appreciable edema.
-
Axial, T2-weighted, short tau inversion recovery (STIR) image through the more distal area of early mineralization reveals prominent soft-tissue edema consistent with early, immature heterotopic ossification.
-
Anteroposterior radiograph performed in external rotation of the right shoulder in a 62-year-old female patient. Amorphous calcification is seen superolateral to the humeral head (arrows). The characteristics of this calcification are not indicative of heterotopic ossification (HO); the mature cortical or trabecular structures seen in mature HO are not found, nor are the well-defined margins that are consistent with the immature woven bone of early HO.




