Practice Essentials
Ependymoma is a central nervous system (CNS) neoplasm composed of glial cells that have differentiated along ependymal lines. These lesions occur most commonly in the ependymal lining of the ventricles, but they also arise in the filum terminale and in the central spinal cord. [1, 2, 3, 4, 5] Ependymoma may arise anywhere within the neuroaxis in children and in adults. The treatment of choice is resection and, in cases of residual disease, adjuvant radiotherapy. [6]
Ependymoma accounts for about 7% of all intracranial neoplasms in adults; in children, ependymoma is the third most common brain tumor. Over 50% of cases occur in children younger than 5 years. [7, 8]
The World Health Organization (WHO) classifies ependymomas as grades I to III, depending on location and histology, as follows: posterior fossa, supratentorial compartment, and spinal cord/canal locations. [7, 8]
(See the image below.)
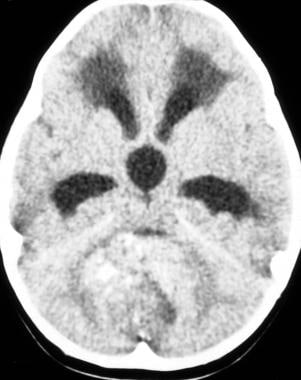 Case 1a. Ependymoma arising from the fourth ventricle. A 13-year-old girl with recent onset of headache, nausea, vomiting, and papilledema. Nonenhanced axial computed tomography image shows a large, round tumor arising within the fourth ventricle with attenuating nodular calcifications. Obstructive hydrocephalus is noted with frontal lobe periventricular white matter low attenuation resulting from subependymal cerebrospinal fluid absorption.
Case 1a. Ependymoma arising from the fourth ventricle. A 13-year-old girl with recent onset of headache, nausea, vomiting, and papilledema. Nonenhanced axial computed tomography image shows a large, round tumor arising within the fourth ventricle with attenuating nodular calcifications. Obstructive hydrocephalus is noted with frontal lobe periventricular white matter low attenuation resulting from subependymal cerebrospinal fluid absorption.
Imaging modalities
Radiologic imaging plays a role in both the diagnostic workup and the treatment of patients with ependymoma; imaging is essential for assessing tumor response to therapy and recurrence. Patients with central nervous system (CNS) symptoms routinely undergo cross-sectional imaging. Computed tomography (CT) scanning is often used initially to evaluate the patient for intracranial hemorrhage, mass, or mass effect. A general limitation of CT is radiation exposure. Additionally, use of iodinated contrast material may sometimes be associated with nausea, vomiting, and rarely anaphylactoid reactions. Limitations of CT with respect to ependymoma include suboptimal anatomic detail.
If a tumor is suspected, magnetic resonance imaging (MRI) is the next study performed. In fact, MRI is the primary imaging modality used in the study of ependymoma. MRI best characterizes CNS tumors, and scan findings should result in an accurate differential diagnosis and frequently in a specific diagnosis. CT scanning is a useful preliminary adjunct, particularly to show tumor calcifications. Before cross-sectional and multiplanar imaging became available, angiography and pneumoencephalography were used to localize brain masses and to characterize tumor vascularity.
General limitations of MRI include cost and the need for patient cooperation. Patient motion is a potential cause of considerable artifact. Patients with claustrophobia and some children require preliminary sedation. Another general limitation of MRI is artifact from metallic foreign bodies and medically implanted objects such as pacemakers. Finally, MRI provides limited benefit in evaluating cortical bone and in detecting calcium.
Ultrasonography, nuclear medicine studies, angiography, myelography/cisternography, and radiography are of limited use in the workup of ependymoma. Fetal ultrasonography and pediatric transcranial sonography are used primarily as screening tools for some pathologic conditions but can detect hydrocephalus reliably.
The final diagnosis of ependymoma, as with most CNS neoplasms, is achieved with tissue biopsy; however, when correlated with demographic and clinical features, MRI and CT scan findings can be strongly suggestive of ependymoma. [9, 10]
Radiography
Radiographic findings are included for historical interest. A study by Barone and Elvidge demonstrated that in 45 pathology proven cases of ependymoma, intracranial calcifications were present in 6 patients. The pineal gland was calcified in 4 patients and was displaced from the midline in 2 patients. Separation of the cranial sutures from raised intracranial pressure occurred in 12 patients. In the 43 patients in whom ventriculography was performed, 41 demonstrated hydrocephalus with identification of the site of obstruction. [11]
Computed Tomography
The characteristics of infratentorial and supratentorial ependymomas are similar on computed tomography (CT) and magnetic resonance imaging (MRI). CT or MRI can be used to evaluate intracranial and spinal ependymomas, but MRI is preferred because it provides enhanced contrast resolution and is better ability to define extent of tumor. However, CT can be useful because it can identify subtle calcifications, which have been reported in up to 50% of ependymomas. Findings of the presence of ependymomas on CT can include hyperdense soft tissue, calcifications, and hemorrhage, and the tumor typically enhances on postcontrast imaging. [7]
Magnetic Resonance Imaging
Magnetic resonance imaging (MRI) is the diagnostic modality of choice in the workup and follow-up of intracranial neoplasms, including ependymoma. The most appropriate role for MRI in the treatment of ependymoma involves detection of tumor and anatomic guidance for surgical resection and/or radiation therapy. MRI is used to monitor ongoing treatment and to search for recurrence. Although MRI findings can be of great help in narrowing the differential diagnosis of brain tumor, final diagnosis is achieved through biopsy with histopathologic analysis.
Solid portions of ependymoma are typically isointense to hypointense relative to white matter on short recovery time/echo time (TR/TE) T1-weighted images. The tumor is hyperintense to white matter on long TR/TE T2-weighted images. As many as 50% of ependymomas reveal signal heterogeneity, which may indicate calcification, necrosis, methemoglobin, hemosiderin, or tumor vascularity. [12, 13, 14] For example, hyperintense foci on both T1- and T2-weighted images suggest methemoglobin in subacute hemorrhage of 1 to 4 weeks' duration, whereas hypointense foci on both T1- and T2-weighted images suggest hemosiderin, calcium, or necrosis.
In the posterior fossa, ependymomas typically arise in the floor of the fourth ventricle and may extend through the foramen of Luschka or the foramen of Magendie. [7]
(See the images below.)
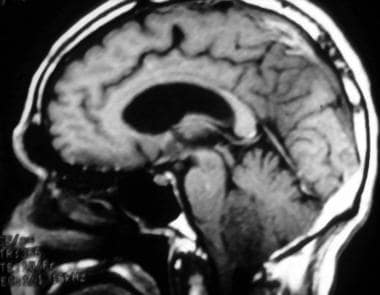 Case 2a. Fourth ventricle ependymoma in a 63-year-old man with headaches. T1-weighted sagittal image shows an oval, fourth ventricular tumor with hypointense signal (likely from tumoral calcifications, confirmed on gradient echo sequence). Moderate obstructive hydrocephalus of the lateral and third ventricles is evident.
Case 2a. Fourth ventricle ependymoma in a 63-year-old man with headaches. T1-weighted sagittal image shows an oval, fourth ventricular tumor with hypointense signal (likely from tumoral calcifications, confirmed on gradient echo sequence). Moderate obstructive hydrocephalus of the lateral and third ventricles is evident.
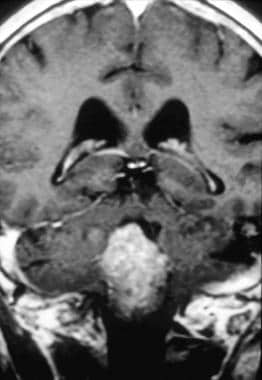 Case 2b. Fourth ventricle ependymoma. T1-weighted coronal postgadolinium intravenous image. Homogeneous enhancement of a fourth ventricular mass is noted, with extension downward through the foramen of Magendie. Pathologic analysis reveals subependymoma.
Case 2b. Fourth ventricle ependymoma. T1-weighted coronal postgadolinium intravenous image. Homogeneous enhancement of a fourth ventricular mass is noted, with extension downward through the foramen of Magendie. Pathologic analysis reveals subependymoma.
Punctate calcific foci are difficult to diagnose prospectively by MRI scan but are present in up to 45% of ependymomas. [14, 15]
(See the images below.)
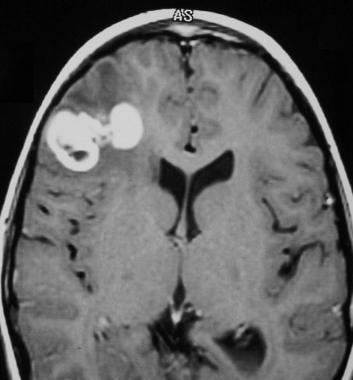 Case 5a. Anaplastic right frontal lobe parenchymal ependymoma in a 5-year-old girl with seizures. Contrast-enhanced T1-weighted axial image shows a heterogeneous mass in the right frontal lobe. Bright contrast enhancement within the neoplasm and areas of low signal intensity are consistent with calcification.
Case 5a. Anaplastic right frontal lobe parenchymal ependymoma in a 5-year-old girl with seizures. Contrast-enhanced T1-weighted axial image shows a heterogeneous mass in the right frontal lobe. Bright contrast enhancement within the neoplasm and areas of low signal intensity are consistent with calcification.
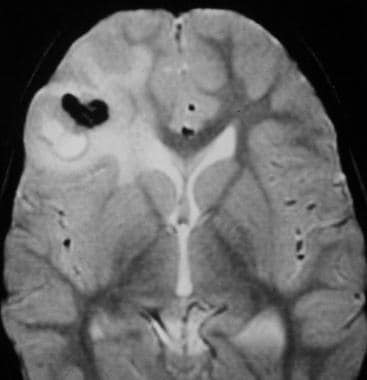 Case 5b. Anaplastic parenchymal ependymoma. T2-weighted axial image shows heterogeneous high signal intensity in the right frontal lobe tumor and adjacent vasogenic edema, with low signal intensity calcifications. No tumor connection to the lateral ventricle is evident on imaging or at the time of surgery. Pathologic analysis shows malignant (anaplastic) ependymoma.
Case 5b. Anaplastic parenchymal ependymoma. T2-weighted axial image shows heterogeneous high signal intensity in the right frontal lobe tumor and adjacent vasogenic edema, with low signal intensity calcifications. No tumor connection to the lateral ventricle is evident on imaging or at the time of surgery. Pathologic analysis shows malignant (anaplastic) ependymoma.
Cystic changes result in high signal intensity on T2-weighted MRI, variably depending on cyst contents.
(See the images below.)
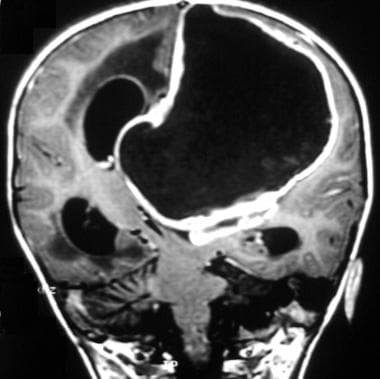 Case 3a. Anaplastic ependymoma of the lateral ventricle in an 8-week-old girl with lateral ventricular hydrocephalus. Gadolinium-enhanced coronal T1-weighted image shows a large anaplastic necrotic ependymoma of the left lateral ventricular roof with severe mass effect and subfalcine herniation.
Case 3a. Anaplastic ependymoma of the lateral ventricle in an 8-week-old girl with lateral ventricular hydrocephalus. Gadolinium-enhanced coronal T1-weighted image shows a large anaplastic necrotic ependymoma of the left lateral ventricular roof with severe mass effect and subfalcine herniation.
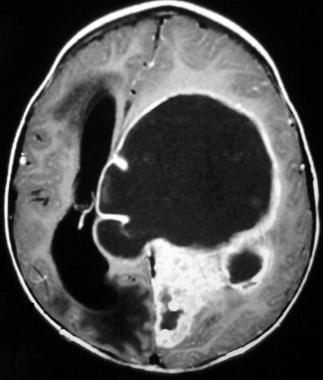 Case 3b. Anaplastic ependymoma of the lateral ventricle. Gadolinium-enhanced axial T1-weighted image shows a large anaplastic ependymoma of the left lateral ventricle with severe mass effect and left-to-right subfalcine herniation.
Case 3b. Anaplastic ependymoma of the lateral ventricle. Gadolinium-enhanced axial T1-weighted image shows a large anaplastic ependymoma of the left lateral ventricle with severe mass effect and left-to-right subfalcine herniation.
Signal heterogeneity is useful in distinguishing ependymoma from the more homogeneous medulloblastoma. Calcification and hemorrhagic foci are more typical of ependymoma than of medulloblastoma. Additionally, an ependymoma is more apt to extend through the foramina of Luschka and Magendie—hence the term “plastic ependymoma.” Similarly, choroid plexus papilloma is more homogeneous than ependymoma and lacks the typical irregular margins of ependymoma.
(See the images below.)
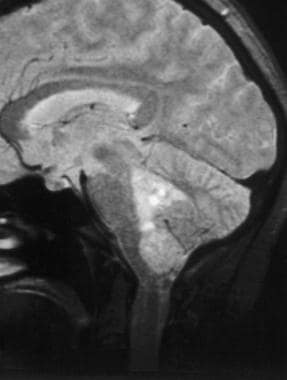 Case 4a. Ependymoma arising from the fourth ventricle in a 50-year-old woman with a history of dizziness and nausea, progressive over several years. The lobulated mass in this proton density–weighted sagittal image arises from the fourth ventricle and extends distally through the foramen of Magendie. Minimal lateral ventricular hydrocephalus is evident. Pathologic analysis reveals cellular ependymoma.
Case 4a. Ependymoma arising from the fourth ventricle in a 50-year-old woman with a history of dizziness and nausea, progressive over several years. The lobulated mass in this proton density–weighted sagittal image arises from the fourth ventricle and extends distally through the foramen of Magendie. Minimal lateral ventricular hydrocephalus is evident. Pathologic analysis reveals cellular ependymoma.
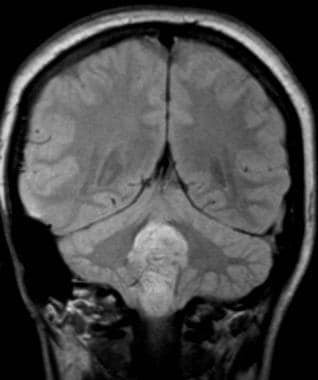 Case 4b. The lobulated fourth ventricular mass in this proton density–weighted coronal image extends distally through the foramen of Magendie. Pathologic analysis reveals cellular ependymoma.
Case 4b. The lobulated fourth ventricular mass in this proton density–weighted coronal image extends distally through the foramen of Magendie. Pathologic analysis reveals cellular ependymoma.
Enhancement with intravenous (IV) gadolinium is useful in differentiating tumor from adjacent vasogenic edema and from normal brain parenchyma. Without IV contrast enhancement, T2-weighted images are more reliable than T1-weighted images in differentiating tumor margins. [15]
Previous cerebral angiography reports describe ependymomas causing displacement of the vein of the lateral recess of the fourth ventricle on cerebral arteriography. [15] This vein normally courses from the transverse and lateral supratonsillar veins along anterior and lateral aspect of the superior pole of the cerebellar tonsil. It then courses lateral to the cerebellopontine angle, over the brachium pontis, to join the petrosal vein. Ependymoma expanding the fourth ventricle and its lateral recesses can displace this vein posteriorly and laterally.
Supratentorial ependymomas (STEs) are more commonly located in the brain parenchyma than infratentorial ependymomas, which are often intraventricular. In addition, STEs are often extraventricular and more often have a cystic component, with or without a mural nodule. In these cases, the differential diagnosis includes ganglioglioma, pilocytic astrocytoma, and pleomorphic xanthoastrocytoma. In the posterior fossa, medulloblastoma and cerebellar astrocytoma can mimic the appearance of an ependymoma. [11]
In a study of 121 patients who had undergone resection followed by radiotherapy for intracracnial ependymomas, Tensaouti et al found that postoperative fluid-attenuated inversion recovery (FLAIR) hyperintensities are a negative prognostic factor. Significant differences were found between patients with postoperative FLAIR abnormalities and those without postoperative FLAIR abnormalities for disease-free survival (71.9% vs. 40.3%; P=0.006) and overall survival (93.7% vs. 72.4%; P= 0.023). [16]
Gadolinium-based contrast agents have been linked to the development of nephrogenic systemic fibrosis (NSF) or nephrogenic fibrosing dermopathy (NFD). NSF/NFD has occurred in patients with moderate to end-stage renal disease after they were given a gadolinium-based contrast agent to enhance MRI or magnetic resonance angiography (MRA) scans. Characteristics include red or dark patches on the skin; burning, itching, swelling, hardening, and tightening of the skin; yellow spots on the whites of the eyes; joint stiffness with trouble moving or straightening the arms, hands, legs, or feet; pain deep in the hip bones or ribs; and muscle weakness. [17]
-
Case 1a. Ependymoma arising from the fourth ventricle. A 13-year-old girl with recent onset of headache, nausea, vomiting, and papilledema. Nonenhanced axial computed tomography image shows a large, round tumor arising within the fourth ventricle with attenuating nodular calcifications. Obstructive hydrocephalus is noted with frontal lobe periventricular white matter low attenuation resulting from subependymal cerebrospinal fluid absorption.
-
Case 1b. Ependymoma of the fourth ventricle. Axial computed tomography image obtained with intravenous contrast agent administration shows dense contrast enhancement in much of the tumor mass and lateral and third ventricular hydrocephalus. Pathologic analysis reveals ependymoma.
-
Case 2a. Fourth ventricle ependymoma in a 63-year-old man with headaches. T1-weighted sagittal image shows an oval, fourth ventricular tumor with hypointense signal (likely from tumoral calcifications, confirmed on gradient echo sequence). Moderate obstructive hydrocephalus of the lateral and third ventricles is evident.
-
Case 2b. Fourth ventricle ependymoma. T1-weighted coronal postgadolinium intravenous image. Homogeneous enhancement of a fourth ventricular mass is noted, with extension downward through the foramen of Magendie. Pathologic analysis reveals subependymoma.
-
Case 3a. Anaplastic ependymoma of the lateral ventricle in an 8-week-old girl with lateral ventricular hydrocephalus. Gadolinium-enhanced coronal T1-weighted image shows a large anaplastic necrotic ependymoma of the left lateral ventricular roof with severe mass effect and subfalcine herniation.
-
Case 3b. Anaplastic ependymoma of the lateral ventricle. Gadolinium-enhanced axial T1-weighted image shows a large anaplastic ependymoma of the left lateral ventricle with severe mass effect and left-to-right subfalcine herniation.
-
Case 4a. Ependymoma arising from the fourth ventricle in a 50-year-old woman with a history of dizziness and nausea, progressive over several years. The lobulated mass in this proton density–weighted sagittal image arises from the fourth ventricle and extends distally through the foramen of Magendie. Minimal lateral ventricular hydrocephalus is evident. Pathologic analysis reveals cellular ependymoma.
-
Case 4b. The lobulated fourth ventricular mass in this proton density–weighted coronal image extends distally through the foramen of Magendie. Pathologic analysis reveals cellular ependymoma.
-
Case 5a. Anaplastic right frontal lobe parenchymal ependymoma in a 5-year-old girl with seizures. Contrast-enhanced T1-weighted axial image shows a heterogeneous mass in the right frontal lobe. Bright contrast enhancement within the neoplasm and areas of low signal intensity are consistent with calcification.
-
Case 5b. Anaplastic parenchymal ependymoma. T2-weighted axial image shows heterogeneous high signal intensity in the right frontal lobe tumor and adjacent vasogenic edema, with low signal intensity calcifications. No tumor connection to the lateral ventricle is evident on imaging or at the time of surgery. Pathologic analysis shows malignant (anaplastic) ependymoma.










