Practice Essentials
Ischemic colitis encompasses a number of clinical entities, all with an end result of insufficient blood supply to a segment or the entire colon. This disease results in ischemic necrosis of varying severity that can range from superficial mucosal involvement to full-thickness transmural necrosis. [1] Bowel ischemia is mainly a disease of old age caused by atheroma of mesenteric vessels. Other causes include embolic disease, vasculitis, fibromuscular hyperplasia, aortic aneurysm, blunt abdominal trauma, disseminated intravascular coagulation, irradiation, and hypovolemic or endotoxic shock. [2] It has been reported in Henoch-Schönlein purpura (HSP), which is one of the collagen vascular diseases. [3] Ischemic colitis is the most prevalent ischemic injury of the GI tract and includes features such as acute abdominal pain, hematochezia,and diarrhea. Colonoscopy along with histopathologic biopsy is the current standard modality for diagnosis. [4, 5]
Venous infarction occurs in young patients, usually after abdominal surgery. [6] Patients may present with colicky abdominal pain, which becomes continuous. It may be associated with vomiting, diarrhea, or rectal bleeding. [7, 8, 9, 10]
Cocaine-induced ischemic colitis is a recognized entity, and the diagnosis is based on clinical and endoscopic findings. Diagnostic imaging is helpful for evaluating abdominal symptoms, and previous studies have suggested specific sonographic findings in ischemic colitis. [11] Elramah et al found that cocaine-related ischemic colitis is associated with a high mortality. The authors’ findings suggest that cocaine-related ischemia should be considered in young patients presenting with acute abdominal pain and/or rectal bleeding with evidence of bowel wall thickening or pneumatosis on either imaging or colonoscopy. The authors went on to propose that testing for cocaine in these patients may help identify patients at high risk of sepsis and death. [12]
Imaging modalities
Abdominal plain radiography is usually an initial examination undertaken in most cases involving acute abdominal problems. Although the initial radiographic findings may be normal in colonic ischemia, it is an invaluable procedure in the differential diagnosis of an acute abdomen. [13] Barium enema results are abnormal in 90% of patients with ischemic colitis. [13, 14, 15, 16, 17]
The radiologic features of ischemic colitis are nonspecific and may be seen in other inflammatory disorders of the colon. Results with all modalities may be normal in patients with established ischemic colitis. A reliable diagnosis of ischemic colitis can be made only when the radiologic findings are correlated with the clinical results.
Abdominal plain film can be used to rule out other emergencies such as organ perforations or intestinal obstructions. The use of barium enema has been almost entirely eplaced by CT imaging or colonoscopy.
CT is the single best test after plain radiography because it can exclude many other causes of abdominal pain and can also establish the diagnosis of intestinal ischemia. For patients who present with symptoms of ischemia, a CT scan of the abdomen with oral and IV contrast and laboratory testing can be performed. [18] Manifestations of ischemic colitis on CT include colonic wall thickening, edema, thumbprinting, bowel dilatation, and effusion of intestinal circumference. Multiphasic CT angiography (CTA) has been recommended for patients with suspected acute mesenteric ischemia or isolated right-sided ischemia. [19, 20]
MRI is mostly useful for magnetic resonance angiography, particularly in individuals with compromised renal function. In addition to MRI being as sensitive as CT in the diagnosis of bowel wall ischemia, it has the added advantage of not requiring ionizing radiation. [21, 22]
Ultrasonography is a noninvasive technique that may provide useful information, particularly in investigating chronic mesenteric ischemia. It is unlikely to be conclusive in excluding mesenteric ischemia. [21, 22, 23] Pneumatosis, a poor prognostic indicator for ischemic colitis, is difficult to identify on US. [24] Ultrasound is sensitive to early structural changes in the colon walls caused by ischemia. Ultrasound can be regarded as an alternative to colonoscopy in patients who are high risk or have contraindications. Positive predictive value of abdominal ultrasound in detecting ischemic colitis has been reported to be 87.5%. A combination of features on ultrasound and clinical findings have been shown to accurately predict outcome and differentiate between mild and severe cases of ischemic colitis. [25, 26]
Angiography has a limited role in cases of colonic ischemia, but it may be invaluable in a few specific indications, such as arteriovenous fistulas and vascular steal syndrome.
(See the images of ischemic colitis below.)
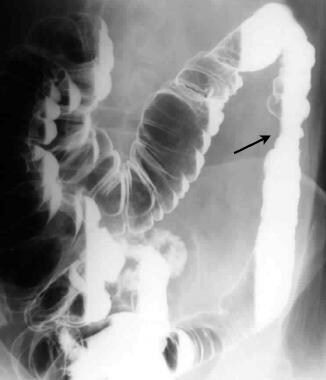 Double-contrast barium enema study shows a stricture of the proximal descending colon secondary to ischemia.
Double-contrast barium enema study shows a stricture of the proximal descending colon secondary to ischemia.
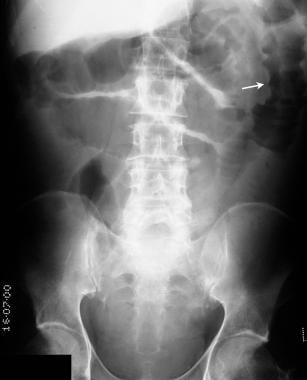 Plain abdominal radiograph in a 65-year-old man presenting with acute abdominal pain and the passage of blood per rectum. Note the thumbprinting in the region of the splenic flexure and also the proximal small-bowel dilatation.
Plain abdominal radiograph in a 65-year-old man presenting with acute abdominal pain and the passage of blood per rectum. Note the thumbprinting in the region of the splenic flexure and also the proximal small-bowel dilatation.
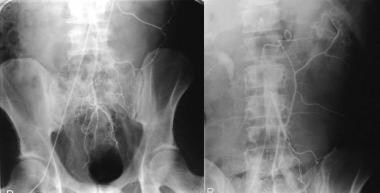
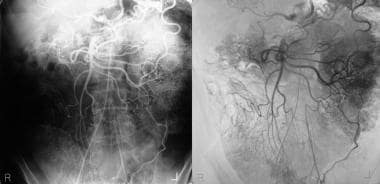 This 63-year-old man had smoked more than 30 cigarettes a day for over 40 years and gave a 3-year history of claudication. Recently, he presented with the passage of mucus and blood per rectum. Flexible sigmoidoscopy revealed a hemorrhagic edematous mucosa with superficial ulceration at the rectosigmoid junction suggestive of ischemic colitis. A flush aortogram showed only the origin of the superior mesenteric artery, but the celiac artery and the inferior artery origins were not identified. A selective superior mesenteric artery angiogram shows retrograde filling of the celiac axis and antegrade filling of the inferior mesenteric artery. Note the arch of Riolan. The image on the right is a manual subtraction image of the angiogram on the left.
This 63-year-old man had smoked more than 30 cigarettes a day for over 40 years and gave a 3-year history of claudication. Recently, he presented with the passage of mucus and blood per rectum. Flexible sigmoidoscopy revealed a hemorrhagic edematous mucosa with superficial ulceration at the rectosigmoid junction suggestive of ischemic colitis. A flush aortogram showed only the origin of the superior mesenteric artery, but the celiac artery and the inferior artery origins were not identified. A selective superior mesenteric artery angiogram shows retrograde filling of the celiac axis and antegrade filling of the inferior mesenteric artery. Note the arch of Riolan. The image on the right is a manual subtraction image of the angiogram on the left.
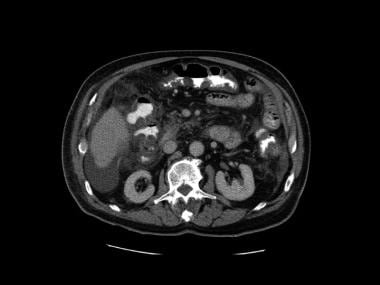
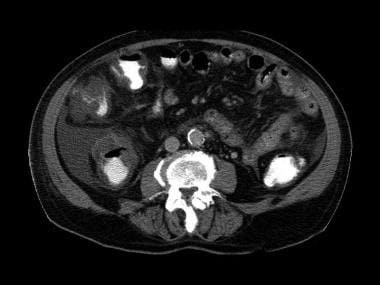
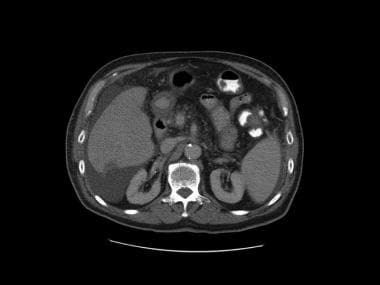
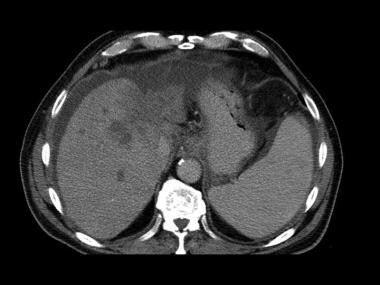
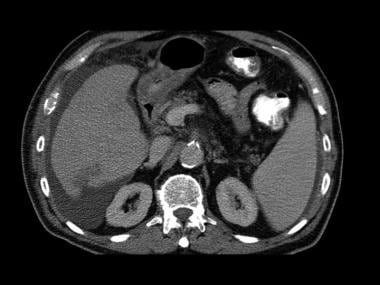
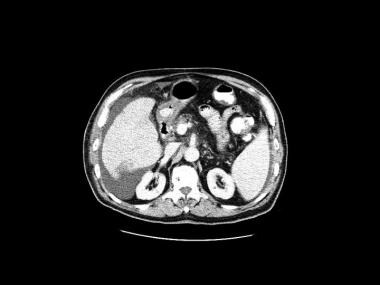
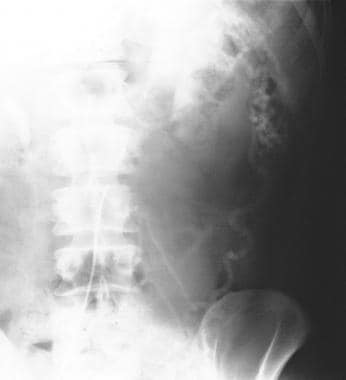
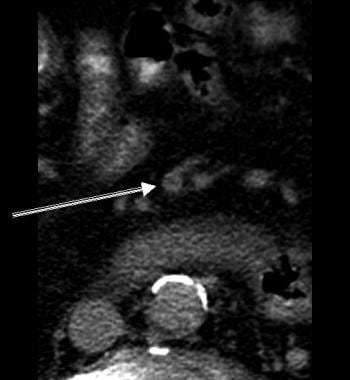 This is the first image in a series of contrast-enhanced CT scans on a 72-year-old man presenting with acute abdominal pain. Surgery revealed mesenteric ischemia, mainly in the right and transverse colon, secondary to peripheral mesenteric thrombosis (arrow). Note the colonic edema akin to thumb printing on plain abdominal radiographs. Multiple hypoattenuating masses in the liver were due to liver abscesses. Moderate ascites is present.
This is the first image in a series of contrast-enhanced CT scans on a 72-year-old man presenting with acute abdominal pain. Surgery revealed mesenteric ischemia, mainly in the right and transverse colon, secondary to peripheral mesenteric thrombosis (arrow). Note the colonic edema akin to thumb printing on plain abdominal radiographs. Multiple hypoattenuating masses in the liver were due to liver abscesses. Moderate ascites is present.
Content.
Radiography
Plain radiographic features include the following:
-
Dilatation of a part of the colon may be seen early. The appearance may be not unlike that of ulcerative colitis with mucosal edema (thumbprinting) and pseudopolyp formation (see the images below).
-
When both the small bowel and the large bowel are affected by ischemic injury, the small bowel may also become dilated with thickening of the valvulae conniventes.
-
Over time, the haustra become thickened and edematous, and it contains a large amount of secretions.
-
With further edema, the haustral markings disappear completely, and the colon acquires a hoselike appearance.
-
Localized pneumatosis coli may be apparent.
-
Eventual tubular narrowing and stricturing appear over a long segment.
 Plain abdominal radiograph in a 65-year-old man presenting with acute abdominal pain and the passage of blood per rectum. Note the thumbprinting in the region of the splenic flexure and also the proximal small-bowel dilatation.
Plain abdominal radiograph in a 65-year-old man presenting with acute abdominal pain and the passage of blood per rectum. Note the thumbprinting in the region of the splenic flexure and also the proximal small-bowel dilatation.
 Left: Plain abdominal radiograph in a 58-year-old man who underwent an upper GI barium study for nonspecific dyspepsia a few days earlier. The patient presented with vague abdominal discomfort and the passage of blood per rectum. Note the thumbprinting in the region of the distal transverse colon and splenic flexure. Right: Another radiograph obtained 24 hours later shows mild dilatation of the distal transverse colon with more obvious mucosal edema. (See also the next image.)
Left: Plain abdominal radiograph in a 58-year-old man who underwent an upper GI barium study for nonspecific dyspepsia a few days earlier. The patient presented with vague abdominal discomfort and the passage of blood per rectum. Note the thumbprinting in the region of the distal transverse colon and splenic flexure. Right: Another radiograph obtained 24 hours later shows mild dilatation of the distal transverse colon with more obvious mucosal edema. (See also the next image.)
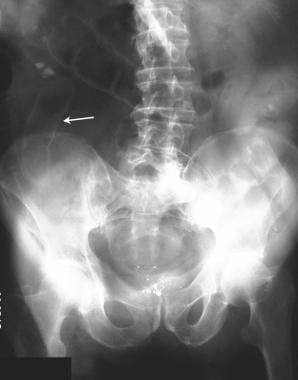 Six days later (see the previous image), the clinical condition of the patient deteriorated, with increasing abdominal pain and signs of peritonism. Note the bowel relief sign. At surgery, a perforated ischemic bowel segment at the splenic flexure was confirmed.
Six days later (see the previous image), the clinical condition of the patient deteriorated, with increasing abdominal pain and signs of peritonism. Note the bowel relief sign. At surgery, a perforated ischemic bowel segment at the splenic flexure was confirmed.
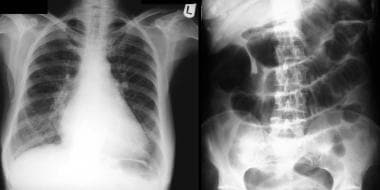 Left: Chest radiograph of a 45-year-old woman with mitral valve disease and atrial fibrillation who presented with acute abdominal symptoms. Note the cardiomegaly and blood diversion in the upper lobe, which is suggestive of cardiac decompensation. Right: Plain radiograph shows several dilated loops of the jejunum. Note also the stricture of the proximal transverse colon. At surgery, a gangrenous small bowel loop secondary to a mesenteric artery embolus was resected. The patient had a similar episode a year earlier, which was managed conservatively; this approach accounts for the ischemic stricture of the proximal transverse colon.
Left: Chest radiograph of a 45-year-old woman with mitral valve disease and atrial fibrillation who presented with acute abdominal symptoms. Note the cardiomegaly and blood diversion in the upper lobe, which is suggestive of cardiac decompensation. Right: Plain radiograph shows several dilated loops of the jejunum. Note also the stricture of the proximal transverse colon. At surgery, a gangrenous small bowel loop secondary to a mesenteric artery embolus was resected. The patient had a similar episode a year earlier, which was managed conservatively; this approach accounts for the ischemic stricture of the proximal transverse colon.
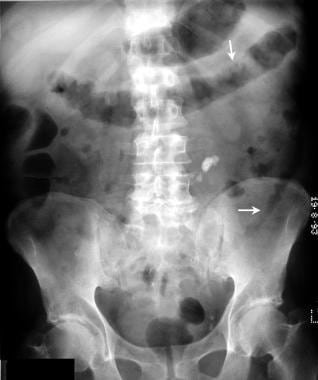 Plain supine abdominal radiograph in a 72-year-old man who presented with bloody diarrhea. Note the thumbprinting of the splenic flexure and the entire descending colon. (See also the next image.)
Plain supine abdominal radiograph in a 72-year-old man who presented with bloody diarrhea. Note the thumbprinting of the splenic flexure and the entire descending colon. (See also the next image.)
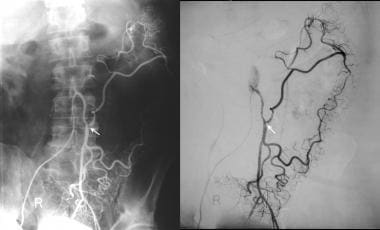 Inferior mesenteric angiogram in the same patient as in the previous image shows a stenosis of more than 50% at the origin of the left colic artery associated with a poststenotic dilatation. Image on the right is a manual subtraction image.
Inferior mesenteric angiogram in the same patient as in the previous image shows a stenosis of more than 50% at the origin of the left colic artery associated with a poststenotic dilatation. Image on the right is a manual subtraction image.
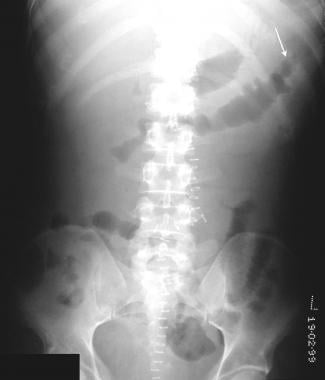 Postoperative abdominal radiograph in a 26-year-old patient who had stabbed himself, cutting across the jejunum. The injury was repaired on an emergency basis. After laparotomy, his recovery was slow, and he had bloody diarrhea. Colonoscopy revealed a hemorrhagic edematous mucosa of the splenic flexure suggestive of ischemic colitis. Note the thumbprinting of the distal transverse colon. (See also the next image).
Postoperative abdominal radiograph in a 26-year-old patient who had stabbed himself, cutting across the jejunum. The injury was repaired on an emergency basis. After laparotomy, his recovery was slow, and he had bloody diarrhea. Colonoscopy revealed a hemorrhagic edematous mucosa of the splenic flexure suggestive of ischemic colitis. Note the thumbprinting of the distal transverse colon. (See also the next image).
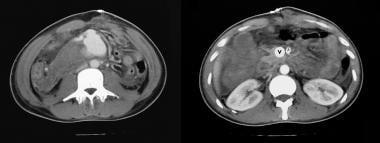 Axial contrast-enhanced CT scans of the same patient as in the previous image shows that the superior mesenteric vein is larger than the aorta. Lower CT section (left) shows a fistula between the superior mesenteric artery and the vein through a pseudoaneurysm, which was the cause of global mesenteric ischemia due to a steal phenomenon. After surgical repair of the arteriovenous communication, the patient fully recovered.
Axial contrast-enhanced CT scans of the same patient as in the previous image shows that the superior mesenteric vein is larger than the aorta. Lower CT section (left) shows a fistula between the superior mesenteric artery and the vein through a pseudoaneurysm, which was the cause of global mesenteric ischemia due to a steal phenomenon. After surgical repair of the arteriovenous communication, the patient fully recovered.
Findings on barium enema examination may include the following:
-
A rapid sequence of events usually occurs over several weeks after an initial ischemic insult. The events can progress from the early stages of spasm and edema to chronic stricture formation.
-
Barium enema examination during the acute stage of a vascular insult demonstrates spasm associated with thickening and blunting of the mucosal folds. Multiple mucosal scalloping or thumbprinting are seen along the contours of the bowel. Seen en face, the thumbprints appear as polypoid filling defects (see the images below).
-
With further progression of mucosal edema, the folds become thickened and ill defined.
-
Intraluminal secretions are increased.
-
Ulcerations are frequently observed in pathologic specimens of the diseased bowel segment. However, these may not be deep enough to be demonstrable radiologically. With diffuse ulceration, the mucosa may be completely effaced. Ulcers may be seen as a serrated mucosa. Deep ulcers are a late finding.
-
During the healing phase, when fibrosis sets in, associated flattening and rigidity of one wall may be observed. The antimesenteric border of the colon becomes pleated because of scarring; this may appear as sacculations or pseudodiverticula.
-
With a continuing fibrotic process, the affected segment of the large bowel acquires a tubular shape with smooth contours and a concentric lumen.
-
The final outcome is usually a long stricture with proximal bowel dilatation. However, when a stricture develops, it is shorter than the original length of the ischemic bowel, as seen radiologically.
-
In the stages before fibrosis develops, partial or complete recovery may occur. The ischemic process may be arrested at any of the previously described stages.
 Double-contrast barium enema study shows a stricture of the proximal descending colon secondary to ischemia.
Double-contrast barium enema study shows a stricture of the proximal descending colon secondary to ischemia.
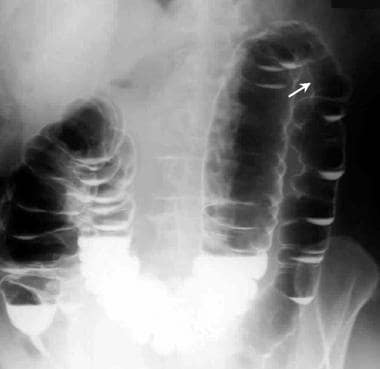 Erect radiograph obtained after a double-contrast barium enema study shows a stricture at the splenic flexure.
Erect radiograph obtained after a double-contrast barium enema study shows a stricture at the splenic flexure.
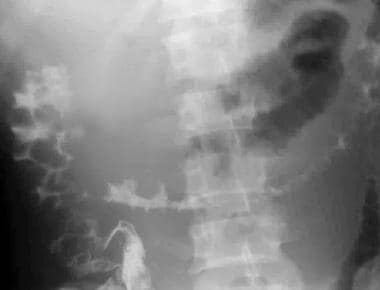 Postevacuation image obtained after a barium enema study shows extensive thumbprinting involving the entire colon, secondary to mesenteric ischemia.
Postevacuation image obtained after a barium enema study shows extensive thumbprinting involving the entire colon, secondary to mesenteric ischemia.
Inflammatory bowel disease, infective colitides, and carcinoma may mimic ischemic colitis. The stricture formation in ischemic colonic disease is smooth and tapering, with a concentric lumen and without shouldering or contour defects.
Sacculations are common in ischemic colitis, but skip lesions are rare. In inflammatory bowel disease, cobblestoning, skip lesions, and small punched-out ulcers are common features. In colonic carcinoma, the narrowing is eccentric and associated with shouldering. In the early stages, contour defects may be observed.
In some cases of ischemic colitis, extensive mucosal ulceration may simulate a lymphosarcoma on a barium enema study.
The plain radiographic findings may be entirely normal, particularly early in the disease. However, the results of barium enema are abnormal in 90% of patients with ischemic colitis.
Computed Tomography
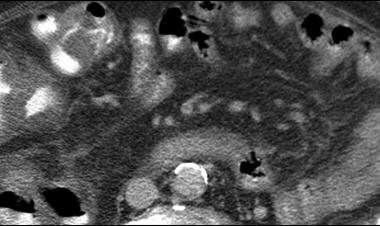
CT has been used as a diagnostic tool in patients with suspected intestinal ischemia. CT not only depicts changes in the blood vessels but also shows changes in the bowel wall, often in association with ancillary abdominal findings. Contrast-enhanced CT also provides information regarding the hemodynamics of blood flow to the bowel (see the images below). [27, 28, 29, 30, 31, 32]
The specific CT signs for establishing the diagnosis of bowel ischemia depend on the degree of ischemia, the length of bowel loops involved, and whether both the large bowel and small bowel are affected. Findings may include the following:
-
Thromboembolism in the mesenteric vessels
-
Intramural or portal venous gas
-
Segmental thickening of the bowel wall
-
Absence of bowel wall enhancement with contrast-enhanced CT
-
Irregular narrowing of the bowel lumen as a result of mucosal edema (thumbprinting)
-
Possible bowel dilatation proximal to the ischemic segment of the bowel
-
Nonspecific signs of bowel ischemia, including bowel obstruction, mesenteric edema, mesenteric vascular engagement, and ascites
 Axial contrast-enhanced CT scans of the same patient as in the previous image shows that the superior mesenteric vein is larger than the aorta. Lower CT section (left) shows a fistula between the superior mesenteric artery and the vein through a pseudoaneurysm, which was the cause of global mesenteric ischemia due to a steal phenomenon. After surgical repair of the arteriovenous communication, the patient fully recovered.
Axial contrast-enhanced CT scans of the same patient as in the previous image shows that the superior mesenteric vein is larger than the aorta. Lower CT section (left) shows a fistula between the superior mesenteric artery and the vein through a pseudoaneurysm, which was the cause of global mesenteric ischemia due to a steal phenomenon. After surgical repair of the arteriovenous communication, the patient fully recovered.
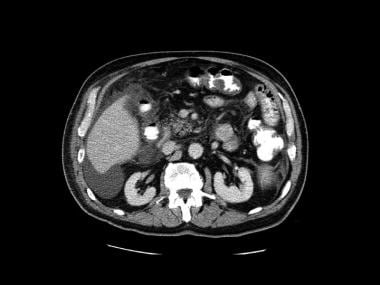
 This is the first image in a series of contrast-enhanced CT scans on a 72-year-old man presenting with acute abdominal pain. Surgery revealed mesenteric ischemia, mainly in the right and transverse colon, secondary to peripheral mesenteric thrombosis (arrow). Note the colonic edema akin to thumb printing on plain abdominal radiographs. Multiple hypoattenuating masses in the liver were due to liver abscesses. Moderate ascites is present.
This is the first image in a series of contrast-enhanced CT scans on a 72-year-old man presenting with acute abdominal pain. Surgery revealed mesenteric ischemia, mainly in the right and transverse colon, secondary to peripheral mesenteric thrombosis (arrow). Note the colonic edema akin to thumb printing on plain abdominal radiographs. Multiple hypoattenuating masses in the liver were due to liver abscesses. Moderate ascites is present.
In a study of patients with symptomatic angiography-verified atherosclerotic obstruction of the superior mesenteric artery (SMA) with acute on chronic mesenteric ischemia (AOCMI) (N = 27) or CMI (N = 20), two thirds of AOCMI patients presented with ischemia-specific CT signs (decreased bowel wall enhancement, pneumatosis, or thrombotic SMA clot). However, the authors noted that any intestinal abnormality in CT together with SMA obstruction should raise suspicion of intestinal ischemia. Most patients with AOCMI had unspecific intestinal findings, such as mesenteric fat stranding in up to 96%, bowel lumen dilatation in 93%, and bowel wall thickening in 70%, while only a few patients with CMI had such findings (due to chronic ischemic colitis). [33]
Degree of confidence
CT is regarded as a valuable noninvasive tool in the diagnosis of mesenteric ischemia. Conflicting reports have appeared in the radiology literature regarding the sensitivity of CT in the diagnosis of mesenteric ischemia. Earlier reports showed a poor sensitivity of around 39%, but more recent reports involving high-quality infusion techniques resulted in a sensitivity of 64-82%. This improved sensitivity is partly related to improved detection rates of more specific signs, such as direct visualization of thromboembolism in the mesenteric vessels and the lack of visualization of bowel wall enhancement.
High-quality images are usually difficult to acquire because of the poor general condition of these patients. CT appears to be significantly limited in detecting small peripheral mesenteric emboli. Some of the well-known CT features of mesenteric ischemia, such as diffuse bowel dilatation, bowel wall thickening, ascites, mesenteric haziness, and vascular engorgement, are elicited in only 36% of the patients. Some of the CT findings are nonspecific and may be found in inflammatory bowel disease.
Magnetic Resonance Imaging
Ha et al showed that the sensitivity of MRI in the detection of bowel ischemia is comparable to that of CT. [21] MRI may be useful in depicting bowel wall changes and in demonstrating mesenteric vascular abnormalities. As with CT, the additional use of contrast enhancement allows an assessment of the dynamic changes in the bowel wall. [22]
In addition to MRI being as sensitive as CT in the diagnosis of bowel wall ischemia, it has the added advantage of not using ionizing radiation. Some researchers have attempted cine phase-contrast MRI for the quantitation of mesenteric blood flow. [23] They measured blood flow in the SMA 30 minutes after the patient ate a meal and found promising criteria for differentiating healthy patients from those with chronic intestinal ischemia.
Mazzei et al demonstrated that unenhanced MRI was adequate for diagnosis and follow-up of many cases of ischemic colitis. [34]
MRI has significant problems in depicting small thromboembolism in small mesenteric vessels.
Ultrasonography
Bowel gas frequently prevents the visualization of colonic changes, which are usually most marked around the splenic flexure. In the initial stages, the ischemic bowel may show increased peristalsis, which is then reduced. The bowel wall becomes thickened, and nodular and intramural hemorrhage and edema give rise to areas of reduced echogenicity. Echogenic areas may be seen in the bowel wall; these may reflect either areas of infarction infiltrate or clot. Echogenic areas with shadowing occur as a result of intramural gas. Gas may also be detected in the portal vein; this is a poor prognostic sign.
Doppler color flow imaging is performed after the patient fasts overnight to minimize bowel gas. The patient is usually scanned in a supine position with the head slightly elevated to allow the abdominal viscera to descend. To identify the celiac axis and the superior mesenteric artery (SMA), the aorta is scanned in the sagittal left paramedian plane, starting at the xiphisternum. The SMA is 1-2 cm distal to the celiac axis; however, the 2 may have a common origin.
Before Doppler studies of the SMA and celiac axis are performed, a Doppler velocity signal in the aorta is obtained at the level of the celiac axis and the SMA at a 60° angle. The aorta is scanned for atheroma, dissection, and aneurysm. Color flow and spectral analysis are used to detect flow disturbances. The peak systolic aortic velocity is recorded, after which velocity measurements are made at the origins of the celiac axis and the SMA.
Care should be taken when recording Doppler time-velocity waveforms at the origins of the celiac axis and the SMA because the insonation angle may be inadvertently increased beyond 60°. Both the celiac axis and the SMA should be examined during both inspiration and expiration to distinguish extrinsic disease (eg, compression of median arcuate ligament of the diaphragm on the celiac axis) from intrinsic disease (atheroma). [35, 36]
Evaluation of the celiac axis
Color flow Doppler sonography is effective in demonstrating flow disturbances associated with tortuosity and stenosis at the origin of the celiac axis. Doppler spectral waveforms in a normal celiac axis after fasting demonstrate a forward flow with an average peak systolic velocity of 123 cm/s ± 9 in persons aged 48-79 years. A significant increase in the systolic or diastolic flow velocity occurs after a meal.
This increase is also reflected in the hepatic and splenic arteries. The average postprandial systolic velocity 30 minutes after a meal of 355 K cal is 132 cm/s ± 7. In the presence of a 60% stenosis of the celiac axis, the peak velocity is increased to 167-208 cm/s ± 9. With this degree of stenosis, color Doppler imaging demonstrates a high-velocity jet at the stenotic site associated with poststenotic turbulent flow.
The potential for collateralization between the celiac axis, SMA, and IMA is remarkable. As a result, the peak systolic velocity in the celiac axis may be lower or much higher than expected when concomitant SMA occlusion is present. This variation may result in the overestimation or underestimation of the extent of ischemic disease.
Evaluation of the SMA
After fasting, the SMA has low diastolic flow. However, after a meal, both the peak systolic and end-diastolic velocities are significantly increased if arterial stenosis is absent. The normal fasting SMA velocities are 128 cm/s ± 16 in persons aged 23-42 years. After a meal, the peak systolic velocity increases to 162 cm/s ± 11; the end-diastolic velocity is in the range of 48 cm/s ± 7 within 15 minutes after the meal. The peak systolic velocity almost doubles within 45 minutes after a meal. With significant SMA stenosis, the peak systolic velocity exceeds 270 cm/s, with a concomitant increase in diastolic flow.
Color flow Doppler imaging shows a jet through the stenotic segment with turbulent flow downstream, which continues for some distance.
Degree of confidence
Ultrasonography is a sensitive yet noninvasive technique that may provide useful information about mesenteric ischemia. Images may demonstrate absent or barely visible color flow, absent arterial signals, and thickened bowel wall loops. Doppler techniques are particularly useful for investigating chronic mesenteric ischemia.
There may be a role for the use of US to assess disease progression in transient ischemic colitis. [24]
Limitations of Doppler analysis of celiac artery and SMA ischemia include the following:
-
There is great potential for collateralization in the splanchnic vessels, which may make an assessment of a single-vessel stenosis difficult
-
The risk of error increases when the angle of insonation is greater than 60°
-
Careful placement of the sample volume is crucial
The SMA must be examined throughout the visualized vessel; otherwise, a false-negative diagnosis may result.
Gas-filled dilated bowel in ileus or small bowel obstruction may prevent adequate visualization of the vasculature.
Nuclear Imaging
Stathaki et al evaluated the use of pentavalent techenetium-99m dimercaptosuccinic acid [Tc-99m (V) DMSA] in the diagnosis of ischemic colitis in 14 patients with endoscopically and histologically confirmed ischemic colitis. Tc-99m (V) DMSA scintigraphy was performed within 2 days after colonoscopy, and images were considered positive when an area of increased activity was observed in the region of interest and considered negative when no abnormal tracer uptake was detected. In 3 of the 14 patients, Tc-99m (V) DMSA images showed moderate activity in the bowel. In the other 11 patients, no abnormal tracer uptake was detected in the abdomen and, therefore, the use of Tc-99m (V) DMSA could not be considered successful in the diagnosis of ischemic colitis. [37]
Angiography
Angiography has a limited role in the diagnosis of colonic ischemia; however, for a few specific indications, angiography may be invaluable. Immediately after an ischemic insult to the colon, the appearance may be normal unless vascular occlusion is present (see the images below). With an increasing colonic inflammatory response, the following may be observed: dilated arteries, prominent accumulation of contrast material in the bowel parenchyma, and dense and early venous filling. The arteriovenous transit time may be accelerated. The venous phase of a superior or inferior mesenteric angiogram may depict small ectatic draining veins. [38]
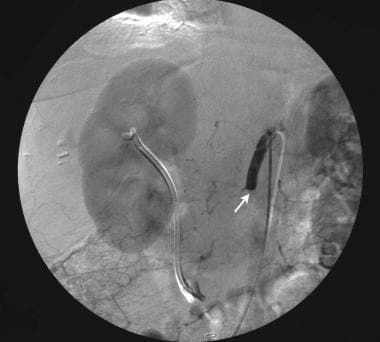 Digital subtracted superior mesenteric angiogram in a 63-year-old woman known to have a hilar cholangiocarcinoma (note the biliary stent) who presented with a sudden onset of abdominal pain and distension. This frame from the angiogram shows a sharp cut-off of the superior mesenteric artery secondary to an acute thrombosis. This patient underwent thrombolysis, with a satisfactory outcome.
Digital subtracted superior mesenteric angiogram in a 63-year-old woman known to have a hilar cholangiocarcinoma (note the biliary stent) who presented with a sudden onset of abdominal pain and distension. This frame from the angiogram shows a sharp cut-off of the superior mesenteric artery secondary to an acute thrombosis. This patient underwent thrombolysis, with a satisfactory outcome.
 This 63-year-old man had smoked more than 30 cigarettes a day for over 40 years and gave a 3-year history of claudication. Recently, he presented with the passage of mucus and blood per rectum. Flexible sigmoidoscopy revealed a hemorrhagic edematous mucosa with superficial ulceration at the rectosigmoid junction suggestive of ischemic colitis. A flush aortogram showed only the origin of the superior mesenteric artery, but the celiac artery and the inferior artery origins were not identified. A selective superior mesenteric artery angiogram shows retrograde filling of the celiac axis and antegrade filling of the inferior mesenteric artery. Note the arch of Riolan. The image on the right is a manual subtraction image of the angiogram on the left.
This 63-year-old man had smoked more than 30 cigarettes a day for over 40 years and gave a 3-year history of claudication. Recently, he presented with the passage of mucus and blood per rectum. Flexible sigmoidoscopy revealed a hemorrhagic edematous mucosa with superficial ulceration at the rectosigmoid junction suggestive of ischemic colitis. A flush aortogram showed only the origin of the superior mesenteric artery, but the celiac artery and the inferior artery origins were not identified. A selective superior mesenteric artery angiogram shows retrograde filling of the celiac axis and antegrade filling of the inferior mesenteric artery. Note the arch of Riolan. The image on the right is a manual subtraction image of the angiogram on the left.
In cases of superior or inferior mesenteric vein thrombosis, the veins may not be visualized, and collateral venous filling may be depicted. Superior or inferior mesenteric arteriovenous fistulas are readily identified with angiography. A vascular steal syndrome is usually diagnosed by means of inferior mesenteric angiography.
In established colonic ischemia, no angiographic changes may be observed, or the angiographic changes may be indistinguishable from those of inflammatory bowel disease. Also, the severity of the ischemic insult and the angiographic changes appear to be poorly correlated.
-
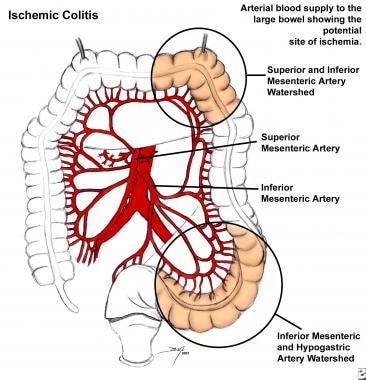
Arterial blood supply to the large bowel shows the potential site of ischemia.
-
Double-contrast barium enema study shows a stricture of the proximal descending colon secondary to ischemia.
-
Erect radiograph obtained after a double-contrast barium enema study shows a stricture at the splenic flexure.
-
Postevacuation image obtained after a barium enema study shows extensive thumbprinting involving the entire colon, secondary to mesenteric ischemia.
-
Plain abdominal radiograph in a 65-year-old man presenting with acute abdominal pain and the passage of blood per rectum. Note the thumbprinting in the region of the splenic flexure and also the proximal small-bowel dilatation.
-
Left: Plain abdominal radiograph in a 58-year-old man who underwent an upper GI barium study for nonspecific dyspepsia a few days earlier. The patient presented with vague abdominal discomfort and the passage of blood per rectum. Note the thumbprinting in the region of the distal transverse colon and splenic flexure. Right: Another radiograph obtained 24 hours later shows mild dilatation of the distal transverse colon with more obvious mucosal edema. (See also the next image.)
-
Six days later (see the previous image), the clinical condition of the patient deteriorated, with increasing abdominal pain and signs of peritonism. Note the bowel relief sign. At surgery, a perforated ischemic bowel segment at the splenic flexure was confirmed.
-
Left: Chest radiograph of a 45-year-old woman with mitral valve disease and atrial fibrillation who presented with acute abdominal symptoms. Note the cardiomegaly and blood diversion in the upper lobe, which is suggestive of cardiac decompensation. Right: Plain radiograph shows several dilated loops of the jejunum. Note also the stricture of the proximal transverse colon. At surgery, a gangrenous small bowel loop secondary to a mesenteric artery embolus was resected. The patient had a similar episode a year earlier, which was managed conservatively; this approach accounts for the ischemic stricture of the proximal transverse colon.
-
Digital subtracted superior mesenteric angiogram in a 63-year-old woman known to have a hilar cholangiocarcinoma (note the biliary stent) who presented with a sudden onset of abdominal pain and distension. This frame from the angiogram shows a sharp cut-off of the superior mesenteric artery secondary to an acute thrombosis. This patient underwent thrombolysis, with a satisfactory outcome.
-
This 63-year-old man had smoked more than 30 cigarettes a day for over 40 years and gave a 3-year history of claudication. Recently, he presented with the passage of mucus and blood per rectum. Flexible sigmoidoscopy revealed a hemorrhagic edematous mucosa with superficial ulceration at the rectosigmoid junction suggestive of ischemic colitis. A flush aortogram showed only the origin of the superior mesenteric artery, but the celiac artery and the inferior artery origins were not identified. A selective superior mesenteric artery angiogram shows retrograde filling of the celiac axis and antegrade filling of the inferior mesenteric artery. Note the arch of Riolan. The image on the right is a manual subtraction image of the angiogram on the left.
-
Normal inferior mesenteric angiogram in a 53-year-old man. (See also the next image.)
-
Normal venous phase of an inferior mesenteric angiogram. (See also the previous image.)
-
Plain supine abdominal radiograph in a 72-year-old man who presented with bloody diarrhea. Note the thumbprinting of the splenic flexure and the entire descending colon. (See also the next image.)
-
Inferior mesenteric angiogram in the same patient as in the previous image shows a stenosis of more than 50% at the origin of the left colic artery associated with a poststenotic dilatation. Image on the right is a manual subtraction image.
-
Postoperative abdominal radiograph in a 26-year-old patient who had stabbed himself, cutting across the jejunum. The injury was repaired on an emergency basis. After laparotomy, his recovery was slow, and he had bloody diarrhea. Colonoscopy revealed a hemorrhagic edematous mucosa of the splenic flexure suggestive of ischemic colitis. Note the thumbprinting of the distal transverse colon. (See also the next image).
-
Axial contrast-enhanced CT scans of the same patient as in the previous image shows that the superior mesenteric vein is larger than the aorta. Lower CT section (left) shows a fistula between the superior mesenteric artery and the vein through a pseudoaneurysm, which was the cause of global mesenteric ischemia due to a steal phenomenon. After surgical repair of the arteriovenous communication, the patient fully recovered.
-
This is the first image in a series of contrast-enhanced CT scans on a 72-year-old man presenting with acute abdominal pain. Surgery revealed mesenteric ischemia, mainly in the right and transverse colon, secondary to peripheral mesenteric thrombosis (arrow). Note the colonic edema akin to thumb printing on plain abdominal radiographs. Multiple hypoattenuating masses in the liver were due to liver abscesses. Moderate ascites is present.
-
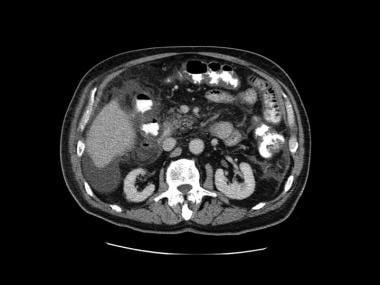
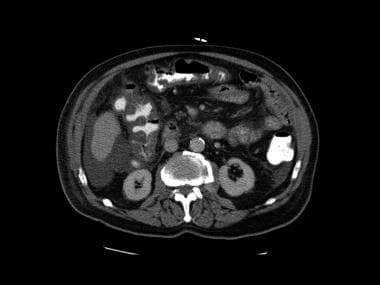
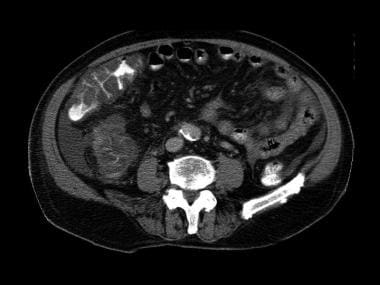
Second image in a series of enhanced CT scans in the same patient.
-
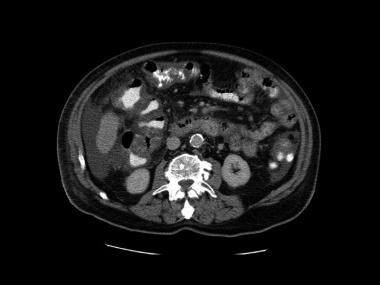
Third image in a series of enhanced CT scans in the same patient.
-
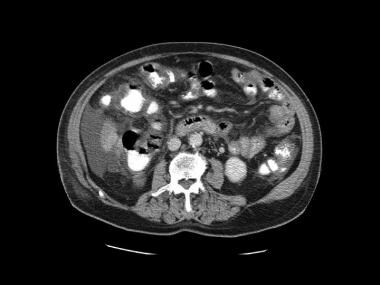
Fourth image in a series of enhanced CT scans in the same patient.
-
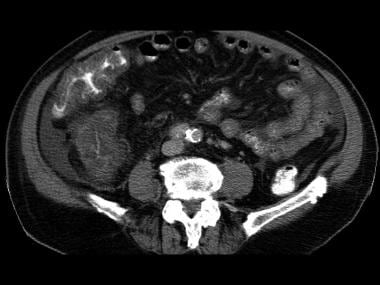
Fifth image in a series of enhanced CT scans in the same patient.
-
Sixth image in a series of enhanced CT scans in the same patient.
-
Seventh image in a series of enhanced CT scans in the same patient.
-
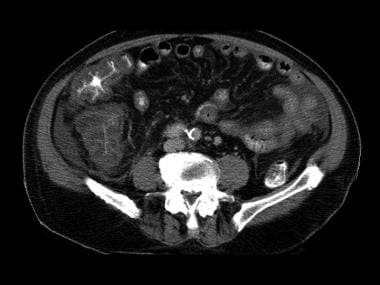
Eighth image in a series of enhanced CT scans in the same patient.
-
Ninth image in a series of enhanced CT scans in the same patient.
-
Tenth image in a series of enhanced CT scans in the same patient.
-
Eleventh image in a series of enhanced CT scans in the same patient.
-
Twelfth image in a series of enhanced CT scans in the same patient.
-
Thirteenth image in a series of enhanced CT scans in the same patient.
-
Fourteenth image in a series of enhanced CT scans in the same patient.
-
Fifteenth image in a series of enhanced CT scans in the same patient.
-
Sixteenth and last image in a series of enhanced CT scans in the same patient.