Practice Essentials
Adrenocortical carcinoma (ACC) (also known as adrenal cortical carcinoma) is a rare malignancy with a poor prognosis, affecting 1-2 people per million per year year. Adrenocortical carcinoma can be seen in familial syndromes such as multiple endocrine neoplasia type 1 (MEN-1), Li-Fraumeni syndrome, Beckwith-Wiedemann syndrome, and Carney complex. An adrenal mass less than 2 cm is likely to be benign. Only 2% of tumors that are less than 4 cm are adrenocortical carcinomas. Masses that are larger than 5 cm are most likely to be malignant. Other features of malignant masses on CT and MRI include irregular edges, heterogeneity, central necrosis, and calcification. [1] The main predictors of ACC recurrence include advanced disease stage, incomplete surgical resection, cortisol production, certain genetic alterations, and high proliferation rate (Ki-67 proliferation index). [2, 3, 1]
Computed tomography (CT), magnetic resonance imaging (MRI), and fluorine-18 fluorodeoxyglucose (FDG) PET/CT are the most common imaging methods for adrenal gland asessment. Well-established morphologic criteria are used for CT and MRI to differentiate between benign and malignant adrenal mass. In cases of known primary malignancy, CT, MRI, and PET can help differentiate most benign masses from metastases. [4, 5, 6, 7, 8, 9] Imaging features indicative of adrenal malignancy are new or enhancing lesions without history of infection, size greater than 4 cm, sudden growth, heterogeneity, margin irregularity, central necrosis, hemorrhage, and calcification. About 30% of cases present as metastatic disease to regional and para-aortic lymph nodes, liver, lung, and bones. [10, 11]
Although CT is used most widely for evaluating abdominal masses, the origin of the mass often is difficult to discern. In addition, the presence or absence of invasion of adjacent structures is difficult to determine in some patients.
Radiomic features derived from preoperative contrast-enhanced CT images have shown encouraging results in predicting the Ki-67 index in patients with ACC. Morphologic features such as shape flatness and elongation have been shown to be superior to other radiomic features in detecting high Ki-67 expression. [12]
(See the images below.)
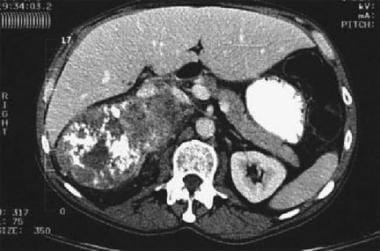
 A 68-year-old woman with a large right upper quadrant primary adrenocortical carcinoma with curvilinear calcification. Low-attenuation regions anteriorly are consistent with necrosis.
A 68-year-old woman with a large right upper quadrant primary adrenocortical carcinoma with curvilinear calcification. Low-attenuation regions anteriorly are consistent with necrosis.
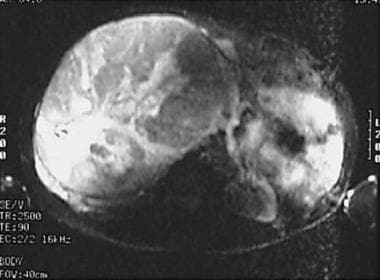 T2-weighted MRI demonstrates a large right adrenocortical carcinoma with high signal intensity involving the right lateral aspect.
T2-weighted MRI demonstrates a large right adrenocortical carcinoma with high signal intensity involving the right lateral aspect.
When identified, tumors frequently are large, measuring 4-10 cm in cross-sectional diameter. Adrenal carcinomas arise from the adrenal cortex and are bilateral in up to 10% of patients. Approximately 50-80% are functional tumors, with most causing Cushing syndrome.
Adrenal incidentalomas are detected in approximately 4-5% of patients undergoing unrelated imaging investigations, such as abdominal CT or MRI. The prevalence of adrenal incidentalomas increases with age, and the majority are identified as benign adrenal adenomas. [13]
In a large patient-population study in the United States, 3982 patients diagnosed with ACC were identified from the National Cancer Data Base (NCDB), and tumor, treatment, and hospital factors associated with survival after resection were examined. Of the patients with nodes examined, 26.5% had nodal metastases, and distant metastases were found in 21.6% of patients. Overall 5-year survival for all patients who underwent resection was 38.6% (median survival, 31.9 months). A higher risk of death was associated with increasing age, poorly differentiated tumors, involved margins, and nodal or distant metastases. [14]
Guidelines
Clinical practice guidelines ACC by the European Society of Endocrinology recommend performing a chest CT in addition to an abdominal-pelvic cross-sectional imaging (CT or MRI) in any case where there is high suspicion for ACC. A detailed hormonal workup is also recommended to identify potential autonomous excess of glucocorticoids, sex hormones, mineralocorticoids, and adrenocortical steroid hormone precursors. Adrenal biopsy is not recommended unless there is evidence of metastatic disease that precludes surgery. [15]
The National Comprehensive Cancer Network (NCCN) guidelines on adrenal tumors finds ACC should be strongly suspected in nonfunctioning tumors greater than 4 cm. CT or MRI scans should be performed using an adrenal protocol to determine size, heterogeneity, lipid content (MRI), contrast washout (CT), and margin characteristics. Chest CT or MRI with contrast of the abdomen and pelvis are also recommended to evaluate for metastatic disease and local invasion when the primary tumor is greater than4 cm. [16]
Radiography
Because adrenal carcinomas are often large at presentation, radiographs of the abdomen may demonstrate mass effect from the tumor. The calcifications observed in more than 30% of patients are often more difficult to detect with abdominal radiographs than with CT scanning.
On excretory urography, adrenal carcinoma often causes mass effect on the ipsilateral superior pole of the kidney, usually displacing the upper pole of the kidney laterally and, when large enough, inferiorly.
With the advent of cross-sectional imaging, the evaluation, staging, and treatment of adrenocortical carcinoma have vastly improved. CT should be the first imaging of choice to define an adrenal mass such as adrenocortical carcinoma.
(See the images below.)
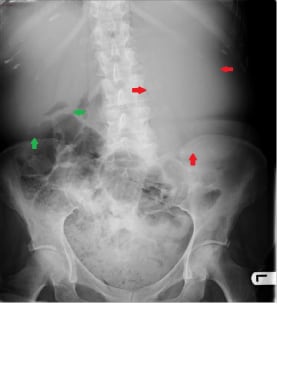 This abdominal radiograph of a 45-year-old patient demonstrates a large soft tissue mass in the left flank (red arrows), which was proven to be adrenal carcinoma subsequently. The mass is pushing the gas-filled bowel loops down. The liver is also enlarged, containing large metastases; its inferior border is reaching the iliac crest (green arrows).
This abdominal radiograph of a 45-year-old patient demonstrates a large soft tissue mass in the left flank (red arrows), which was proven to be adrenal carcinoma subsequently. The mass is pushing the gas-filled bowel loops down. The liver is also enlarged, containing large metastases; its inferior border is reaching the iliac crest (green arrows).
Computed Tomography
On CT scans, adrenocortical carcinoma appears as a large mass, often with central necrosis. Calcification, seen in the images below, is observed in as many as 30% of patients.
 A 68-year-old woman with a large right upper quadrant primary adrenocortical carcinoma with curvilinear calcification. Low-attenuation regions anteriorly are consistent with necrosis.
A 68-year-old woman with a large right upper quadrant primary adrenocortical carcinoma with curvilinear calcification. Low-attenuation regions anteriorly are consistent with necrosis.
On unenhanced images, heterogeneity is often found with larger masses. On enhanced images, the tumor enhances heterogeneously, with the greatest enhancement often at the periphery and often irregular.
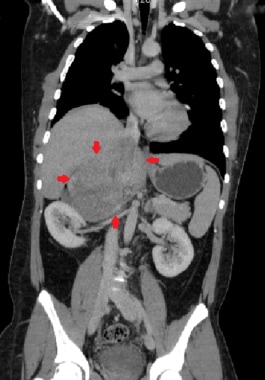 This coronal CT reconstruction demonstrates a large heterogeneously enhancing right adrenal carcinoma (red arrows).
This coronal CT reconstruction demonstrates a large heterogeneously enhancing right adrenal carcinoma (red arrows).
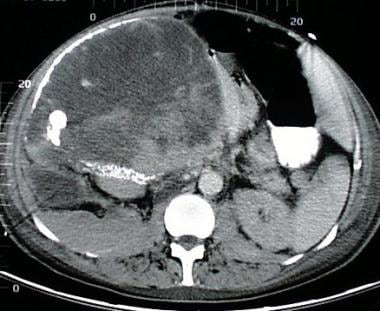
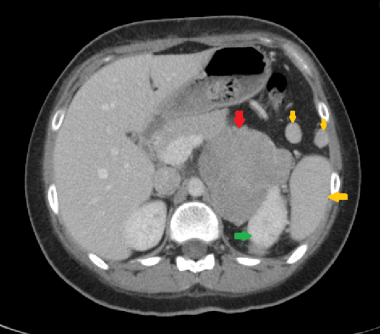 This axial CT shows a large left adrenal carcinoma (red arrow). It abuts the left kidney (green arrow) without any definite evidence of direct invasion. Spleen (large yellow arrow) and two splenunculi (small yellow arrows) are also shown (the latter were also present on a previous CT scan from 5 years ago and were morphologically unchanged).
This axial CT shows a large left adrenal carcinoma (red arrow). It abuts the left kidney (green arrow) without any definite evidence of direct invasion. Spleen (large yellow arrow) and two splenunculi (small yellow arrows) are also shown (the latter were also present on a previous CT scan from 5 years ago and were morphologically unchanged).
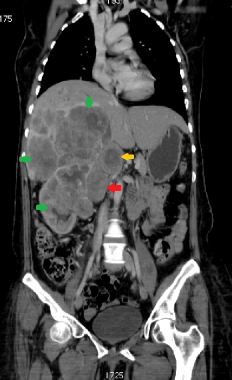 This coronal CT reconstruction shows right adrenal carcinoma (red arrow) with an adjacent involved node (yellow arrow). Extensive direct invasion of right kidney and liver is also seen (green arrows).
This coronal CT reconstruction shows right adrenal carcinoma (red arrow) with an adjacent involved node (yellow arrow). Extensive direct invasion of right kidney and liver is also seen (green arrows).
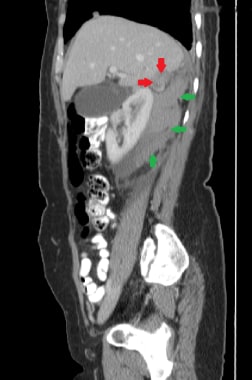 This 28-year-old patient presents with acute hemorrhage (green arrows) secondary to a right adrenal carcinoma (red arrows), as shown in this sagittal CT reconstruction.
This 28-year-old patient presents with acute hemorrhage (green arrows) secondary to a right adrenal carcinoma (red arrows), as shown in this sagittal CT reconstruction.
CT findings that increase the index of suspicion for adrenocortical carcinoma include the following:
-
Large mass (>4 cm)
-
Central necrosis or hemorrhage
-
Heterogeneous enhancement
-
Invasion into adjacent structures
-
Venous extension into the renal vein or inferior vena cava
Occasionally, differentiating an adrenal carcinoma from other pathology in the upper abdomen may be difficult because the mass is large and the fat planes are indistinct. In these patients, multiplanar magnetic resonance imaging (MRI) is the better imaging test. In particular, the imaging findings of large pheochromocytomas and metastasis may be identical. Therefore, it may be prudent to obtain spot vanillylmandelic acid (VMA) or metanephrines prior to resection to prevent a hyperintensive crisis as not all pheochromocytomas are clinically overt.
False-positive lesions could include exophytic renal masses and exophytic pancreatic tail masses.
Radiomic features derived from preoperative contrast-enhanced CT images have shown encouraging results in predicting the Ki-67 index in patients with ACC. Morphologic features such as shape flatness and elongation have been shown to be superior to other radiomic features in detecting high Ki-67 expression. [12]
Magnetic Resonance Imaging
MRI often demonstrates a large mass with lower signal intensity than the liver on T1-weighted images and higher signal intensity than the liver on T2-weighted images. Often, the tumor demonstrates heterogeneous hyperintensity on T1- and T2-weighted images because of the central necrosis and hemorrhage. Because the mass usually does not contain any significant intracellular lipid, it will not lose signal on out-of-phase imaging.
MRI is advantageous for evaluating tumors, since its depiction of vascular invasion and extension into surrounding structures often is superior to that of CT. Additionally, the most cephalad extension of the tumor must be evaluated so that the surgeon can obtain vascular control of the tumor. This can be achieved with CT but often is easier with MRI.
Larger adrenal adenomas are radiologically similar to adrenal cortical carcinomas. The pathologic distinction between adrenal adenoma and adrenal carcinoma is largely based on size, with the cutoff in the range of 4-5 cm.
(See the images below.)
 T2-weighted MRI demonstrates a large right adrenocortical carcinoma with high signal intensity involving the right lateral aspect.
T2-weighted MRI demonstrates a large right adrenocortical carcinoma with high signal intensity involving the right lateral aspect.
Coronal and sagittal images may be helpful in determining adrenal origin of the mass, thus differentiating it from renal cell carcinoma or hepatocellular carcinoma, especially if CT is equivocal.
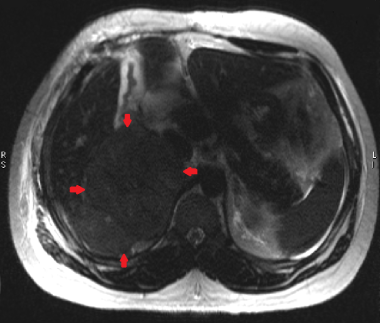 This axial T2W HASTE image demonstrates a large right adrenal carcinoma (red arrows), which is slightly hyperintense compared to the liver.
This axial T2W HASTE image demonstrates a large right adrenal carcinoma (red arrows), which is slightly hyperintense compared to the liver.
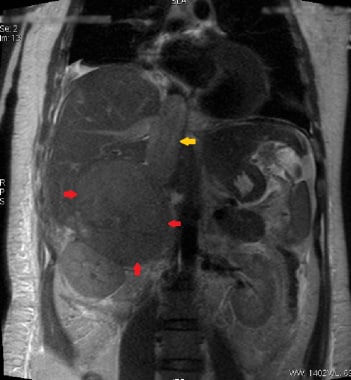 This coronal T2W HASTE image shows a large right adrenal carcinoma (red arrows) invading into the inferior vena cava (yellow arrow).
This coronal T2W HASTE image shows a large right adrenal carcinoma (red arrows) invading into the inferior vena cava (yellow arrow).
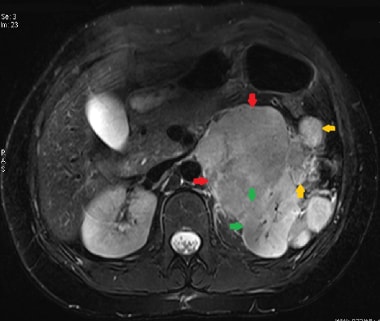 Axial T2W fat-saturated image demonstrates a hyperintense large left adrenal carcinoma (red arrows). There is evidence of direct invasion of the left kidney (green arrows). A few adjacent metastases are also seen (yellow arrows).
Axial T2W fat-saturated image demonstrates a hyperintense large left adrenal carcinoma (red arrows). There is evidence of direct invasion of the left kidney (green arrows). A few adjacent metastases are also seen (yellow arrows).
Ultrasonography
Adrenocortical carcinoma demonstrates a homogeneous echo texture when small, but the echo pattern becomes heterogeneous with cystic areas when the tumor grows as a result of hemorrhage and necrosis.
(See the images below.)
Because different planes are obtainable on ultrasonography, it is helpful in some patients to determine the organ of origin of the mass.
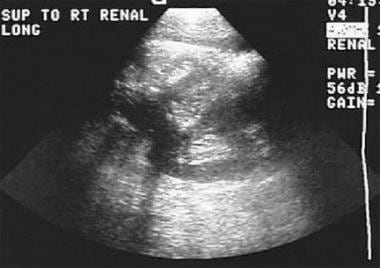
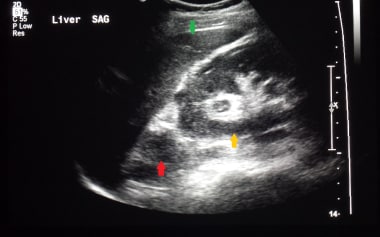 A case of early right adrenal carcinoma (red arrow). The image also shows a normal liver (green arrow) and right kidney (yellow arrow).
A case of early right adrenal carcinoma (red arrow). The image also shows a normal liver (green arrow) and right kidney (yellow arrow).
Nuclear Imaging
Nuclear scintigraphy does not play much of a role in the evaluation of adrenal carcinoma, except to exclude other lesions such as pheochromocytomas or aldosteronomas.
Iodine-121 metaiodobenzylguanidine (MIBG) and indium-111 octreotide can be used to visualize pheochromocytomas, while iodine-131 6-beta-iodomethyl-19-norcholesterol (NP-59) can be used to detect aldosteronomas or other hyperfunctioning cortical tumors. [17, 18]
Positron emission tomography imaging performed with fluorine-18 fluorodeoxyglucose (FDG) has shown promise in differentiating benign adrenal lesions from malignant lesions. [19] FDG PET/CT, as compared to contrast-enhanced CT (CECT), can better diagnose adrenal malignancies and has better prognostic outcome. [20] PET/CT is preferred for chemotherapeutic response assessment, as it may predict response before anatomic changes are detected on CT. [21] FDG PET/CT has higher specificity to rule out suspected adrenocortical carcinoma (ACC) recurrences diagnosed by CT. Therefore, FDG PET/CT can play a significant role in patient management as a second-line modality in the postoperative surveillance of ACC patients after CT detection of a potential recurrence. [22]
(See the images below.)
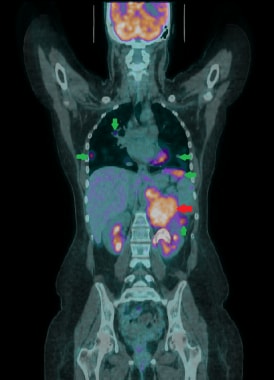 Coronal fused 18F-FDG PET/CT image of a 27-year-old male. This staging scan shows a moderately FDG avid large left adrenal carcinoma (red arrow) with multiple peritoneal and lung metastases (green arrows).
Coronal fused 18F-FDG PET/CT image of a 27-year-old male. This staging scan shows a moderately FDG avid large left adrenal carcinoma (red arrow) with multiple peritoneal and lung metastases (green arrows).
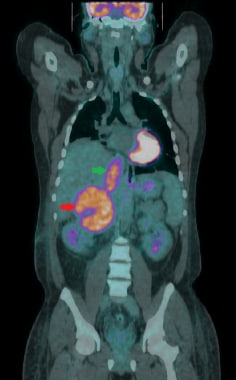 Coronal fused 18F-FDG PET/CT image showing a large FDG avid right adrenal carcinoma (red arrow), which is invading into the inferior vena cava (green arrow).
Coronal fused 18F-FDG PET/CT image showing a large FDG avid right adrenal carcinoma (red arrow), which is invading into the inferior vena cava (green arrow).
Angiography
Prior to cross-sectional imaging, arteriography was the preferred modality for evaluating abdominal masses. On angiograms, adrenal carcinomas are usually hypovascular masses, which helps distinguish them from hypernephromas. Little vascular shunting, puddling, or venous laking is found with adrenal carcinoma compared with renal cell carcinoma. Usually, faint tumor vascularity is seen on abdominal aortograms, and it is not until selective adrenal arteriography is performed that tumor vessels are identified. The predominant arterial supply to the adrenal gland and to adrenal carcinoma is the superior adrenal artery off the inferior phrenic artery.
-
A 68-year-old woman with a large right upper quadrant primary adrenocortical carcinoma with curvilinear calcification. Low-attenuation regions anteriorly are consistent with necrosis.
-
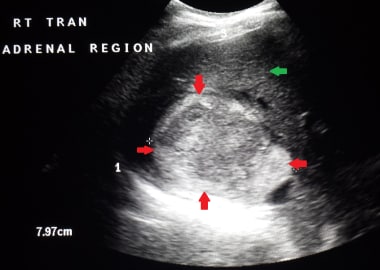
A 67-year-old man with a heterogeneous mass superior to the right kidney.
-
CT demonstrates a large heterogeneous mass, with flocculent calcifications and central necrosis.
-
T2-weighted MRI demonstrates a large right adrenocortical carcinoma with high signal intensity involving the right lateral aspect.
-
This abdominal radiograph of a 45-year-old patient demonstrates a large soft tissue mass in the left flank (red arrows), which was proven to be adrenal carcinoma subsequently. The mass is pushing the gas-filled bowel loops down. The liver is also enlarged, containing large metastases; its inferior border is reaching the iliac crest (green arrows).
-
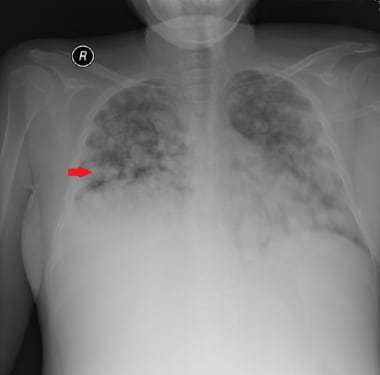
This 3- year-old patient presented with advanced-stage adrenal carcinoma. Numerous bilateral nodular lung metastases can be seen (red arrow).
-
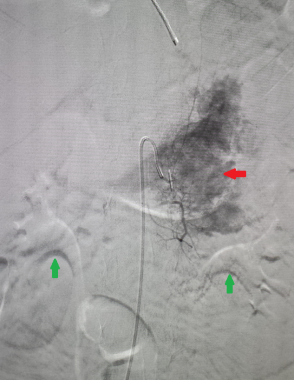
This selective angiogram shows "tumor blush" within a large left adrenal carcinoma (red arrow). Bilateral renal pelvicalyceal systems are also seen (green arrows).
-
A case of early right adrenal carcinoma (red arrow). The image also shows a normal liver (green arrow) and right kidney (yellow arrow).
-
This image shows a relatively large right adrenal carcinoma (red arrows) abutting the liver (green arrow).
-
This coronal CT reconstruction demonstrates a large heterogeneously enhancing right adrenal carcinoma (red arrows).
-
This axial CT shows a large left adrenal carcinoma (red arrow). It abuts the left kidney (green arrow) without any definite evidence of direct invasion. Spleen (large yellow arrow) and two splenunculi (small yellow arrows) are also shown (the latter were also present on a previous CT scan from 5 years ago and were morphologically unchanged).
-
This coronal CT reconstruction shows right adrenal carcinoma (red arrow) with an adjacent involved node (yellow arrow). Extensive direct invasion of right kidney and liver is also seen (green arrows).
-
This 28-year-old patient presents with acute hemorrhage (green arrows) secondary to a right adrenal carcinoma (red arrows), as shown in this sagittal CT reconstruction.
-
This axial T2W HASTE image demonstrates a large right adrenal carcinoma (red arrows), which is slightly hyperintense compared to the liver.
-
This coronal T2W HASTE image shows a large right adrenal carcinoma (red arrows) invading into the inferior vena cava (yellow arrow).
-
Axial T2W fat-saturated image demonstrates a hyperintense large left adrenal carcinoma (red arrows). There is evidence of direct invasion of the left kidney (green arrows). A few adjacent metastases are also seen (yellow arrows).
-
Coronal fused 18F-FDG PET/CT image of a 27-year-old male. This staging scan shows a moderately FDG avid large left adrenal carcinoma (red arrow) with multiple peritoneal and lung metastases (green arrows).
-
Coronal fused 18F-FDG PET/CT image showing a large FDG avid right adrenal carcinoma (red arrow), which is invading into the inferior vena cava (green arrow).
-
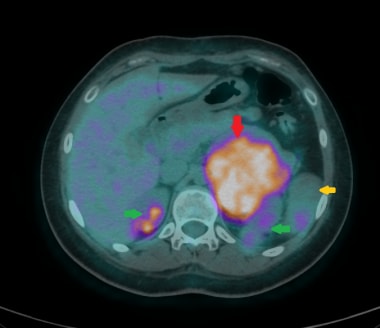
This axial fused 18F-FDG PET/CT image shows a large markedly FDG avid left adrenal carcinoma (red arrow). Physiologic uptake within both kidneys is seen (green arrows). Spleen (yellow arrow) is partially visualized.